Guide B-719
Jason L. Turner
College of Agricultural, Consumer and Environmental Sciences, New Mexico State University
Extension Horse Specialist, Department of Extension Animal Sciences and Natural Resources, New Mexico State University. (Print Friendly PDF)
Introduction
West Nile virus (WNV) was first reported in Uganda, Africa, in 1937. Before arriving in Long Island, New York, in 1999, it was widely distributed in Africa, the Middle East, southwest Asia, and parts of Europe. Since 2000, WNV has spread across the continental U.S., and cases in equines have been reported in every state except Hawaii and Alaska.
The virus causes encephalomyelitis, or inflammation of the brain and spinal cord, in equine animals. While passerine birds serve as the primary reservoir for the virus, natural or experimental infection, with or without clinical signs of disease, has been documented in a variety of mammals and reptiles. There is great concern for WNV as a zoonotic disease of humans. Most infected people do not show any signs of illness or only develop mild symptoms like fever, head and body aches, nausea, and vomiting. However, immunosuppressed people, children, and the elderly may develop neurological symptoms of the disease, which can be fatal.

Hunter Folsom on Unsplash.
Clinical Signs
While all horses are susceptible to WNV disease, many horses infected with WNV may not show clinical symptoms, and some horses can die without showing signs of disease prior to death. The incubation period in horses (the time from infection with WNV to onset of clinical symptoms) is three to 15 days.
The initial symptoms include signs of discomfort or anxiety, lameness, fever, loss of appetite, and depression. As the disease progresses, neurological symptoms develop, which include muzzle twitching, impaired vision, inability to swallow, head pressing, irregular gait and incoordination, trembling, muscle contractions, paralysis, convulsions, weakness, and death. Horses may also become hypersensitive to sights, sounds, and touch.
The fatality rate for horses that show clinical symptoms of WNV can be as high as 50%. Horses that do recover typically begin to show signs of improvement within a week after the onset of clinical symptoms. While many horses will recover from the disease, as many as 40% of those infected may show long-lasting defects, such as changes in behavior, muscle atrophy, weakness in one or more limbs, and reduced exercise tolerance.
Disease Transmission
Wild passerine birds are the primary vertebrate reservoir hosts for WNV, and the degree and duration of viral replication varies with the species of bird. WNV infection has been documented in over 320 species of birds in North America since 1999. Some species carry the virus without showing clinical symptoms of disease, while others will develop the disease. It is believed that migratory bird populations are important in introducing WNV into previously unaffected regions of the U.S.
In North America, more than 60 species of mosquitoes have been shown to harbor WNV. These mosquitoes become infected by feeding on infected birds, and then transmit the virus when they bite an equine or human (Figure 1). Both equines and humans are considered “dead-end” hosts because they may develop the disease, but they do not have a high enough degree of virus replication sufficient to transmit the virus to other mammals or mosquitoes.
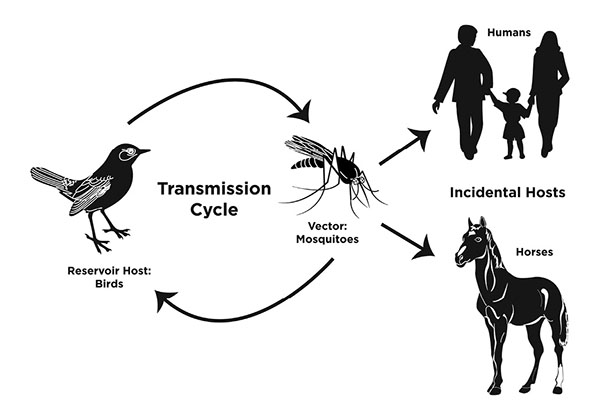
Figure 1. The transmission cycle for West Nile virus in horses (adapted from Long, 2019).
Outbreaks of WNV in equines are most prevalent during the summer and early fall, and the transmission cycle normally ends when freezing temperatures eliminate infected mosquito populations. Although the process is not well understood, the virus may overwinter, in dormant mosquitoes that survive the winter or in birds, to reintroduce the virus in the following year when environmental conditions favor transmission.
Prevention
Since there is no treatment for WNV (other than supportive care), it is best to prevent disease occurrence through vaccination. Since 2003, the use of licensed vaccines that are commercially available in the U.S. has greatly reduced the incidence of WNV infection in equines (Figure 2). In the unlikely instance where a vaccinated horse contracts the disease, the severity of the disease and the chances for recovery are significantly better than for unvaccinated horses.
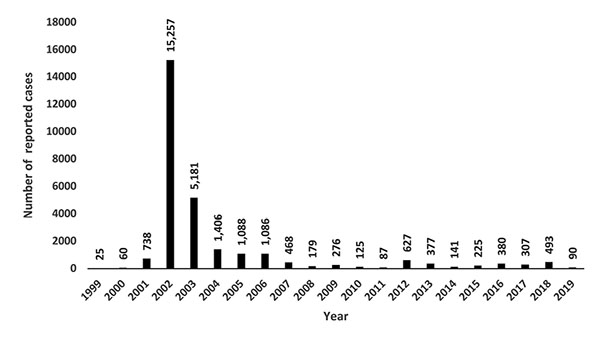
Figure 2. The number of reported equine cases of West Nile virus infection in the U.S., 1999–2019. (Source: Data from the Animal and Plant Health Inspection Service, United States Department of Agriculture.)
It is recommended to administer the horse’s annual booster vaccination just prior to the start of “mosquito season” in your area. In areas of the country where the long mosquito season may outlast the duration of protective immunity provided by annual vaccination, horses may require more frequent booster vaccinations to maintain adequate protection. It is important that equine owners consult their veterinarian to develop a vaccination program for WNV and other mosquito-borne diseases that fits their individual needs and risk of exposure. The core vaccination guidelines of the American Association of Equine Practitioners (www.aaep.org) provide some basic recommendations as a starting point.
Other preventive measures that minimize the risk of exposure to mosquitoes that transmit WNV include:
- Reduce standing water, which provides an area where mosquitoes can reproduce. This includes draining water from any item that can hold water (e.g., old tires, flower pots, children’s toys, etc.). Tanks or other water sources for animals should be cleaned at least once per week to prevent mosquito growth. When the water source can’t be “cleaned,” using an approved larvicide, such as Bacillus thuringiensis israelensis (Bti), can reduce mosquito numbers.
- Reduce areas that may provide cover for adult mosquitoes by mowing weeds and tall plants.
- Using screens, fans, or insecticide fogging systems for stabled horses can reduce their exposure. To further minimize the risk of exposure, coordinate riding or turn-out times outside of peak mosquito activity, which is normally around dusk and dawn, and avoid turning on lights in the stables during the evening or overnight period.
- In non-stabled horses, fly masks, fly sheets, and insecticides or mosquito repellents are effective. Be sure to follow manufacturer label directions for the specific product used.
- Owners should also protect themselves against mosquitoes by wearing clothing that covers their skin, such as long sleeve shirts or gloves, while wearing a DEET-containing repellent, and avoiding the outdoors during peak mosquito activity times.
Conclusion
Equine owners should understand the severe consequences of WNV in their equines as well as the risk for disease in humans. Therefore, it is important that responsible owners use the effective WNV vaccines available along with other mosquito control practices to minimize the risk of their horse developing the fatal disease.
References
American Association of Equine Practitioners. n.d. Core vaccination guidelines [Online]. https://aaep.org/guidelines/vaccination-guidelines/core-vaccination-guidelines
American Association of Equine Practitioners. 2018. 10 tips for reducing your horse’s West Nile risk [Online]. https://aaep.org/horsehealth/10-tips-reducing-your-horses-west-nile-risk
Animal and Plant Health Inspection Service, United States Department of Agriculture. n.d. West Nile virus (WNV), distribution maps [Online]. https://www.aphis.usda.gov/aphis/ourfocus/animalhealth/animal-disease-information/equine/wnv/west-nile-virus
Hansen, I.A., et al. 2017. Stop those suckers [Infographic; online].
Long. M.T. 2007. Flavivirus infections. In D.C. Sellon and M.T. Long (Eds.), Equine Infectious Diseases, 1st ed. (pp. 198–206). St. Louis: Saunders Elsevier.
Long, M.T. 2019. West Nile encephalomyelitis in horses [Online]. The Merck veterinary manual. https://www.merckvetmanual.com/horse-owners/brain,-spinal-cord,-and-nerve-disorders-of-horses/west-nile-encephalomyelitis-in-horses
Merck & Co., Inc. 2020. The Merck veterinary manual [Online]. https://www.merckvetmanual.com
Spickler, A.R. 2013. West Nile virus infection [Online]. https://www.cfsph.iastate.edu/Factsheets/pdfs/west_nile_fever.pdf
United States Environmental Protection Agency. 2017. Bti for mosquito control [Online]. https://www.epa.gov/mosquitocontrol/bti-mosquito-control
For Further Reading
B-708: Documents Required to Transport Horses in New Mexico
https://pubs.nmsu.edu/_b/B708/
Guía B-708: Documentos Requeridos para el Transporte de Caballos en Nuevo México
https://pubs.nmsu.edu/_b/B708sp/
B-716: Equine Herpesvirus in Horses
https://pubs.nmsu.edu/_b/B716/
B-717: Vesicular Stomatitis Virus in Horses
https://pubs.nmsu.edu/_b/B717/
B-718: Eastern Equine Encephalitis in Horses
https://pubs.nmsu.edu/_b/B718/

Jason L. Turner is Associate Professor and Extension Horse Specialist. Jason was active in 4-H and FFA while growing up in Northeastern Oklahoma. His M.S. and Ph.D. studies concentrated on equine reproduction, health, and management. His Extension programs focus on proper care and management of the horse for youth and adults.
To find more resources for your business, home, or family, visit the College of Agricultural, Consumer and Environmental Sciences on the World Wide Web at pubs.nmsu.edu.
Contents of publications may be freely reproduced, with an appropriate citation, for educational purposes. All other rights reserved. For permission to use publications for other purposes, contact pubs@nmsu.edu or the authors listed on the publication.
New Mexico State University is an equal opportunity/affirmative action employer and educator. NMSU and the U.S. Department of Agriculture cooperating.
July 2020 Las Cruces, NM
