Guide H-658
Richard Heerema
College of Agricultural, Consumer and Environmental Sciences, New Mexico State University
Author: Extension Pecan Specialist, Department of Extension Plant Sciences, New Mexico State University. (Print Friendly PDF)
Introduction
Nutrient deficiencies and toxicities are among the most important limiting factors for productivity in commercial and backyard pecan orchards in New Mexico. Fourteen mineral elements are considered essential for plants to grow and produce flowers and fruit. Six of these—nitrogen, phosphorous, potassium, calcium, magnesium, and sulfur—are classified as macronutrients because they are normally found in the plant tissues in relatively high concentrations. Depending on the particular element and plant tissue, macronutrient concentrations normally range from about 0.1 to 3.0% of tissue dry weight (Table 1). The remaining eight nutrients—zinc, manganese, iron, copper, boron, nickel, molybdenum, and chlorine—are classified as micronutrients. They are normally found in far lower concentrations, a few hundred parts per million down to 1 or fewer parts per million (ppm) of tissue dry weight, again depending on the element and tissue (Table 1).
Table 1. "Adequate" Concentration Range of Macronutrients and Micronutrients Recommended for Leaf Tissue of New Mexico Pecan Trees Sampled Late July to Early August
|
Macronutrients (%)
|
|||||||
|
Nitrogen (N)
|
Phosphorus (P)
|
Potassium (K)
|
Calcium (Ca)
|
Magnesium (Mg)
|
Sulfur (S)
|
||
|
2.5—3.0
|
0.14—0.19
|
1.2—2.5
|
0.9—1.8
|
0.3—0.6
|
0.15—0.35
|
||
|
Micronutrients (ppm or mg per kg)
|
|||||||
|
Zinc (Zn)
|
Manganese (Mn)
|
Iron (Fe)
|
Copper (Cu)
|
Boron (B)
|
Nickel (Ni)
|
Molybdenum (Mo)
|
Chlorine (Cl)
|
|
50—100
|
100—300
|
50—250
|
8—30
|
50—150
|
>2.5
|
>2.0(?)
|
N/A
|
Pecan trees have a minimum threshold content of each nutrient required in order for growth, flowering, and nut production to proceed optimally. When tissue concentrations of a nutrient drop below that threshold, pecan orchard performance declines and the trees are said to be deficient for that nutrient (Figure 1). Likewise, orchard performance may be negatively affected when the content of a nutrient is excessive, reaching toxic levels in the tissue. Mild nutrient deficiencies may go unnoticed by orchardists because, although nut production or shoot growth is negatively affected, the trees exhibit no obvious visible deficiency symptoms. This is called hidden hunger. With increasingly severe deficiencies, orchard performance worsens and visible nutrient deficiency symptoms may begin to appear.
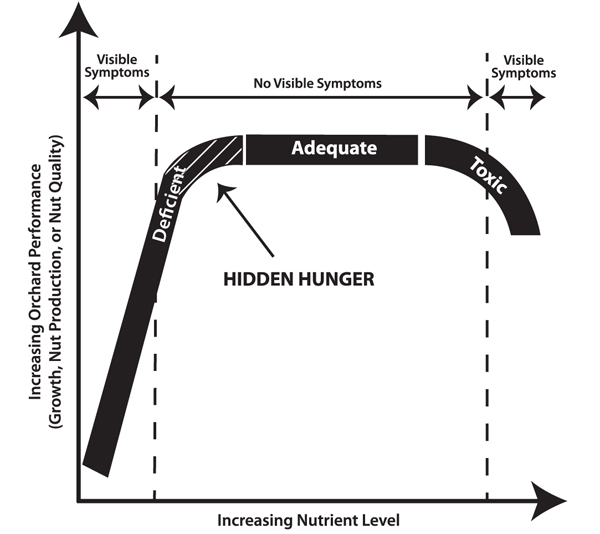
Figure 1. Pecan trees may experience nutrient deficiency or toxicity without exhibiting any visible symptoms.
Many of the essential nutrients have characteristic deficiency or toxicity symptoms that are valuable to pecan growers for identifying and diagnosing nutrient imbalances and spotting problem areas in orchard blocks. To maintain orchard nutrition for optimum orchard performance, growers should be familiar with nutrient deficiency and toxicity symptoms in pecan (symptoms may differ from other plant species) and closely monitor their pecan orchards throughout the season for these symptoms. When evaluating nutrient deficiency symptoms, remember that trees may exhibit multiple nutrient deficiency symptoms at the same time!
Visible symptoms alone, however, are not sufficient for monitoring pecan orchard nutritional status because orchard performance typically declines before any visible symptoms appear. Pecan growers should annually collect leaf tissue samples for nutrient analyses to monitor orchard nutrient status trends and to develop a fertilizer program that maintains orchard nutrient levels within an adequate range over the long term. Periodic analysis of soil samples from orchards can also help producers evaluate the underlying reasons behind particular nutritional imbalances in an orchard.
Pecan leaf tissue sampling method
The most important thing to remember when sampling pecan leaflets for tissue nutrient analyses is to use a consistent method. If your leaflet sampling method varies, you will not be able to make season-to-season or block-to-block comparisons and will miss the opportunity to spot important nutritional trends in your orchard blocks over a period of several years.
A uniform leaf sampling protocol has been developed that allows pecan growers to compare leaf tissue nutrient concentrations with those of New Mexico State University's published nutrient concentration ranges (Table 1).
For regular annual leaf tissue nutrient analyses, the recommended sampling procedure for pecans is to sample the middle pair of leaflets from the compound leaf in the middle of the current season's shoot (Figure 2) in late July or early August. Collect leaflets from all sides of the tree that are exposed to sunlight and can be easily reached from the ground (in some orchards you may need to stand in a truck bed or on a short ladder). If possible, avoid sampling leaflets with wind or insect damage, evidence of disease, or other abnormalities.
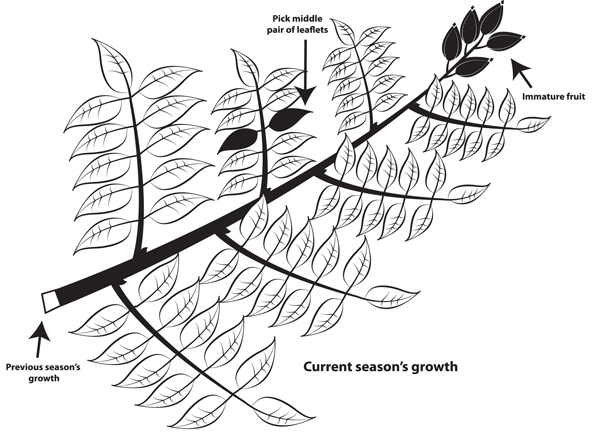
Figure 2. Recommended location to collect leaflet samples for tissue nutrient analyses.
The concentration of nutrients in leaflet tissue varies with sampling time in the season, position of the leaf on the shoot, and position of the leaflet in the leaf, so it is not valid to compare nutrient analysis results to NMSU's tissue nutrient concentration values if your leaflet tissue was sampled using a different protocol.
To diagnose a nutrient imbalance symptom or production issue in a problem area of the orchard, it may sometimes be useful to sample leaflets at a different time or in some other way not in accordance with the described protocol. In such special cases, sample leaves from both the problem trees and from healthy trees on the same day, and using the same method for both. This will give you a healthy or "normal" nutrient concentration to use for comparison in diagnosing the problem area.
For routine leaf sampling, collect leaflets from each 10-acre or smaller orchard block for analysis as a separate sample. Collect a minimum of 60 leaflets per block, sampling from representative trees over the entire area. If there are different cultivars, extreme differences in soil type, or other obvious environmental zones within an orchard block, separately sample each unique area and each cultivar in the block. For an additional layer of consistency, some growers collect leaflet samples from the same marked rows or trees every year.
To avoid contaminating samples after collection, do not expose them to galvanized metals (which contain zinc), soil, fertilizers, or phosphate-containing detergents. If leaflets have been sprayed in the current season with foliar micronutrient fertilizers, external fertilizer residues must be washed off. Wash leaflets with 0.1% hydrochloric acid solution in distilled water, then rinse three times with distilled water. In orchards where there have been no foliar micronutrient sprays in the current season, leaflets may be simply washed in tap water and a small amount of phosphate-free detergent and then rinsed three times with distilled water. Leaflets to be analyzed only for macronutrients do not need be washed.
Once the leaflets have been washed, blot them dry, place them in a perforated paper bag (to allow airflow), and air-dry them at room temperature. Put the dry leaf samples in fully labeled (date, orchard name, sample number) paper bags. Seal the paper bags with tape and ship to an analytical laboratory for nutrient analysis.
Nutrient descriptions
Macronutrients
Nitrogen (N). Nitrogen is part of the structure of many important biochemicals, including proteins, DNA, RNA, chlorophyll, and some plant hormones (e.g., auxins and cytokinins). Nitrogen nutrition is closely tied to canopy rates of photosynthesis because much of the N in pecan trees is part of the photosynthetic apparatus in the leaves. It appears that bearing trees have two periods of especially high N demand: 1) during the flush of vegetative growth at the beginning of the growing season (late April through mid-June) and 2) during the kernel fill stage at the end of the season (September through October, depending on cultivar). Nitrogen must be applied annually in most pecan orchards because pecan trees have relatively high N demand, and N is only present transiently in soils (due to leaching and gaseous losses). Nitrogen-deficient pecan trees exhibit general leaf chlorosis, poor vegetative vigor, low nut quality, and small, poorly filled nuts. The leaves of N-deficient pecan trees often begin fall senescence during kernel fill. Conversely, pecan trees with excessive N levels may be overly vegetative (and have less than optimal nut production) and have increased susceptibility to insect pests. Excessive N nutrition in young non-bearing pecan orchards may make trees more susceptible to injury from early fall freezes. Recommended leaf N concentration in July to August is 2.5 to 3.0%.
Phosphorus (P). Phosphorus is a component of DNA and RNA molecules and the lipids that form cell membranes. Phosphorus also forms high-energy chemical bonds with sugars and adenosine molecules for storing and transferring energy within and between plant cells. In calcareous soils with high pH, P availability is reduced, but visible P deficiency symptoms are rare in New Mexico pecans. Recommended leaf P concentration in July to August is 0.14 to 0.19%.
Potassium (K). Potassium is involved in regulating the amount of water and ions inside plant cells and within the different parts of plant cells. One notable example is the opening and closing of the stomates (also known as stomata), the openings on the undersurface of pecan leaves that allow CO2 from the atmosphere to enter the leaf for photosynthesis, but also allow loss to the atmosphere of water vapor from the leaf (transpiration). The pair of guard cells that surround each stomate are able to change their shape by rapidly adjusting their K concentration. This allows pecan stomates to open and close in response to changing environmental stimuli, such as light level, so that CO2 acquisition is maximized but water loss is minimized. Potassium also activates numerous enzymes that are essential to plant function. Developing pecan fruit, especially the shuck tissues, have a very high K demand. Much (or all) of the K that growing pecan fruit accumulate is supplied by reallocation from nearby leaves in the second half of the growing season when the kernel is developing and accumulating oil and protein (Smith, 2009; Diver and Smith, 1984); thus, it is critical for leaves to contain adequate K early in the season. Recommended leaf K concentration in July to August is 1.2 to 2.5%.
Calcium (Ca). Calcium interacts with pectin to help strengthen cell walls and also activates a few enzymes, including some that are involved in responses to salinity and other stresses. The protein calmodulin activates many different enzymes but must be bound to four Ca2+ ions in order to achieve its proper and active shape. Calcium is involved in stomatal control, pollen tube growth, and root hair growth. Even without the application of Ca-containing fertilizers, Ca deficiency is unlikely to occur in New Mexico because most soils naturally contain adequate supplies of Ca (in the form of lime) to meet tree uptake demands. Recommended leaf Ca concentration in July to August is 0.9 to 1.8%.
Magnesium (Mg). Magnesium is the central atom of the chlorophyll molecule, which serves to capture light energy for photosynthesis; thus, it is not surprising that photosynthesis is negatively affected by Mg deficiency. Magnesium activates numerous enzymes, including enzymes involved in constructing RNA polymers, protein synthesis, and energy transfer in the cell. Magnesium deficiency is characterized by interveinal chlorosis (yellowing or bronzing in the area between the leaf veins, while the area around the veins maintains a greener color) or necrosis. Older leaves are typically affected before younger leaves. Marginal leaf scorch may occur in trees with severe deficiencies. Magnesium deficiency is common in pecan orchards in the southeastern U.S. where soils are often acidic, but are rare in New Mexico's alkaline pH soils. Magnesium deficiencies may occasionally occur in New Mexico in orchards with excessive soil Ca or K. Recommended leaf tissue Mg concentration in July to August is 0.3 to 0.6%.
Sulfur (S). Three of the twenty standard amino acids (the basic sub-units of all proteins, including enzymes) contain S in their molecular structure. The presence and location of these amino acids in a protein determine its shape, structure, and function. Sulfur deficiencies are unlikely in New Mexico pecan orchards. Recommended leaf tissue S concentration in July to August is 0.15 to 0.35%.
Micronutrients
Zinc (Zn). Pecan trees have a higher Zn requirement than most other fruit and nut trees. Zinc availability for root uptake is low in soils with pH above 7.0, which is the norm in New Mexico pecan orchards. Thus, virtually all New Mexico pecan orchards require foliar applications of Zn during the early part of each season while leaf expansion and shoot growth are underway. Orchards with courser-textured soil (sand or sandy loam) are especially prone to Zn deficiencies. Zinc is poorly mobile within the pecan tree, so good Zn spray coverage in the canopy is critical, and repeated applications (3—5 for mature orchards, 5—8 for immature orchards) are usually necessary. 'Wichita' cultivar pecan trees are far more prone to Zn deficiency than other popular New Mexico cultivars such as 'Western' ('Western Schley'). Zinc is an important component or activator of numerous plant proteins, including proteins involved in DNA synthesis, photosynthesis, and root tolerance of flooded soils. Adequate Zn nutrition is necessary for normal metabolism of the auxin hormone indoleacetic acid (IAA), which regulates shoot and leaf growth patterns among many other things. The usual visible symptoms of Zn deficiency are shortened internodes (the part of the stem between leaf nodes) and small leaves with interveinal chlorosis and rippled leaf margins (Figures 3 and 4). This suite of symptoms is called "rosette" and "little leaf." In severely Zn-deficient trees, interveinal necrosis and shoot dieback eventually become evident. Oftentimes, the symptom form and severity in Zn-deficient pecan orchards vary greatly among trees and even among branches within the same tree canopy. Zinc deficiency greatly inhibits growth and development of young pecan trees and nut production in mature orchards. Producers should also be cautious not to over-supply their orchards with Zn because excess Zn can create imbalances with other micronutrients such as nickel. Excess N, P, and Mn may contribute to Zn deficiency issues in pecan orchards. Recommended leaf Zn concentration in July to August is 50 to 100 ppm. Remember that for tissue Zn analyses to provide a meaningful number, leaves should be acid-washed to remove foliar spray residues before shipping to the laboratory.

Figure 3. Pecan leaves showing rippled leaf margins characteristic of zinc deficiency.

Figure 4. Pecan leaves showing signs of zinc deficiency.
Manganese (Mn). Manganese is best known for its role as part of the light-energy-capturing reactions of photosynthesis in which water molecules are split and oxygen gas is released. The energy from these reactions is ultimately stored in carbohydrates, food for growth and production of the pecan tree. Visible Mn deficiency symptoms include general leaf chlorosis or interveinal leaf chlorosis. While tissue Mn levels are usually low in New Mexico pecan orchards, they are usually not so low that Mn deficiency symptoms appear. Orchards may benefit from one or two foliar Mn sprays each spring or early summer. In pecan orchards with acidic soils, which are extremely rare in the Southwest, Mn toxicity symptoms (late budbreak, small leaves, shortened internodes) have occurred when Mn accumulated in leaf tissues in concentrations exceeding 4,000 ppm (Nuñez, 2009). Recommended leaf Mn concentration in July to August is 100 to 300 ppm.
Iron (Fe). Iron is a component of several molecules that help carry out photosynthesis and is also involved in biosynthesis of chlorophyll. Thus, photosynthesis is heavily dependent on Fe nutrition. Visible Fe deficiency symptoms include severe interveinal chlorosis in leaves that still have normal shape and size (Figure 5). In the worst cases, the chlorotic leaf tissues may be very light yellow to almost white in color and, over time, branch and limb dieback can occur if left untreated (Figure 6). Compared to Zn, Fe deficiency symptom severity is even more variable tree-to-tree in an orchard and branch-to-branch in a single tree canopy. Iron deficiency symptoms occur regularly in pecan orchards with very high lime content (10—25%), as in the Pecos Basin. In orchards with very high lime content, Fe deficiency is best treated with soil application of Fe-EDDHA chelated fertilizers or foliar applications of chelated Fe fertilizers. Most pecan-growing areas in New Mexico, such as the Mesilla Valley, have more moderate lime content (<10%). When Fe deficiency symptoms occur in pecan trees growing on moderate-lime-content soils, it is likely caused by excessive irrigation (especially flood irrigation), excessive rainfall, or poor soil water drainage. Correcting the drainage or overwatering issues usually will eliminate the Fe deficiency symptoms. Recommended leaf Fe concentration in July to August is 50 to 250 ppm, but be aware that the connection between leaf nutrient concentration and deficiency symptom severity is not as strong for Fe as it is for other nutrients.

Figure 5. Pecan leaf with severe interveinal chlorosis typical of iron deficiency.

Figure 6. Pecan tree that has experienced branch and limb dieback due to iron deficiency.
Copper (Cu). Copper, like many of the other micronutrients, activates or is contained in many different enzymes and proteins, including several involved in photosynthesis and cellular respiration. Copper deficiency is not well described in pecan, but Cu is similar to other previously discussed metal micronutrients in that it is poorly available for root uptake from alkaline and calcareous soils. Recommended leaf Cu concentration in July to August is 8 to 30 ppm.
Boron (B). Boron, like Ca, functions primarily in cell wall synthesis, stability, and structure. Reproductive function, in particular pollination and fertilization, is particularly sensitive to B nutrition. In general, although B is more readily available in acidic than alkaline pH soils, B deficiencies are far less common in New Mexico pecans than B toxicities. Visible B toxicity symptoms include marginal leaf scorch or necrosis (identical in appearance to salinity injury). Boron toxicities are typically associated with high B concentrations (e.g., >2 ppm) in irrigation well water. Recommended leaf B concentration in July to August is 50 to 150 ppm.
Nickel (Ni). Nickel activates the enzyme urease, which is involved in converting N transported from storage tissues in the trunk, branches, and roots in the spring into a usable form in the growing shoots and expanding leaves. Nickel deficiency symptoms include sparse springtime foliage, slow budbreak, blunted leaflet tips ("mouse ear"), and brittle wood. In New Mexico, Ni deficiencies occur most commonly in orchards with young trees, those located in colder growing areas (e.g., Valencia and Quay Counties), and those with very sandy or high-lime-content soils. Nickel deficiency may be amended with foliar Ni fertilizer application in the fall or spring. Recommended leaf Ni concentration is greater than 2.5 ppm.
Molybdenum (Mo). Molybdenum is required by plants for proper N metabolism. In contrast to the other micronutrients, Mo availability is lowest in acid soils. Availability of Mo in most New Mexico soils is therefore expected to be good. Mo deficiency or toxicity has not been documented
in pecan.
Chlorine (Cl). Like Mn, Cl is essential as part of the water-splitting/oxygen-evolving complex in the photosynthetic light reactions. Chlorine deficiencies have not been documented in pecans, but Cl toxicities do occur. Chlorine toxicity symptoms are identical in appearance to B toxicity symptoms.
References and further reading
Barker, A.V., and D.J. Pilbeam. 2007. Handbook of plant nutrition. New York: Taylor and Francis Group.
Diver, S.G., and M.W. Smith. 1984. Influence of fruit development on seasonal elemental concentrations and distribution in fruit and leaves of pecan. Communications in Soil Science and Plant Analysis, 15, 619—637.
Nuñez Moreno, J.H. 2009. Nutritional studies on pecans [Carya illinoinensis (Wangenh.) C. Koch] growing in irrigated alkaline soils [Ph.D. dissertation]. Tucson: University of Arizona.
Pond, A.P., J.L. Walworth, M.W. Kilby, R.D. Gibson, R.E. Call, and H. Nuñez. 2006. Leaf nutrient levels for pecans. HortScience, 41, 1339—1341.
Smith, M.W. 2009. Partitioning phosphorus and potassium in pecan trees during high- and low-crop seasons. Journal of the American Society for Horticultural Science, 134, 399—404.
Wells, L. (Ed.). 2007. Cultural management of commercial pecan orchards. In Southeastern pecan growers' handbook (pp. 61—82). Athens: University of Georgia Cooperative Extension Service.

Richard Heerema is the Extension pecan specialist. He earned his Ph.D. in plant biology from the University of California—Davis. His research includes micronutrient nutrition, irrigation efficiency, rootstocks, and alternate bearing in pecan. His Extension work provides New Mexico's growing pecan industry with research-based information and tools needed to maximize profitability and sustainability.
To find more resources for your business, home, or family, visit the College of Agricultural, Consumer and Environmental Sciences on the World Wide Web at pubs.nmsu.edu
Contents of publications may be freely reproduced for educational purposes. All other rights reserved. For permission to use publications for other purposes, contact pubs@nmsu.edu or the authors listed on the publication.
New Mexico State University is an equal opportunity/affirmative action employer and educator. NMSU and the U.S. Department of Agriculture cooperating.
July 2013 Las Cruces, NM