CR 650
Robert Flynn, April Ulery, William C. Lindemann
College of Agricultural, Consumer and Environmental Sciences, New Mexico State University
Authors: Respectively, Extension Agronomy Specialist, Department of Extension Plant Sciences; Soil Scientist, Department of Plant and Environmental Sciences; and Soil Microbiologist, Department of Plant and Environmental Sciences, all of New Mexico State University. (Print Friendly PDF)
Overview of Sulfur in Plant Nutrition
Sulfur (S) is an essential nutrient for plant growth. It is considered a macronutrient and must be available in relatively large amounts for good crop growth—much like nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), and magnesium (Mg). Sulfur is present in plants as part of the amino acids cysteine, cystine, and methionine, which make proteins. These amino acids account for about 90% of the S in plants. Thus, protein synthesis and photosynthetic rates are decreased when S is deficient. Cysteine and methionine are also precursors of other S-containing compounds, such as coenzymes and secondary plant products. Sulfur is part of the structure of these compounds or acts as a functional group directly involved in metabolic reactions. Plants mainly absorb S in the form of sulfate ions (SO4-2) (Mengel and Kirkby, 1982).
Nationwide, deficiencies of S in crops are thought to be increasing (McGrath and Zhao, 1995). Sulfur deficiencies are attributed to improved fertilizers that contain little to no S impurities, intensive cropping systems that leave behind little organic matter, increased yield that results in more S removal, less deposition of S from the atmosphere, and less use of S-containing pesticides.
Sulfur-deficient soils are often low in organic matter, coarse textured, well drained, and subject to leaching because sulfate is mobile in the soil. In semi-arid regions, SO4-2 can accumulate in the lower soil profile as soluble gypsum. The S status of New Mexico's soils is not well defined, and S effects on the growth of New Mexico crops have not been extensively researched.
Plant Sulfur Needs
Sulfur deficiency symptoms are sometimes difficult to distinguish from N deficiency. However, in contrast to N deficiency, S deficiency symptoms first occur in the younger, most recently developed leaves. Plants that are S-deficient have a reduced rate of growth and can be rigid and brittle, and the stems remain relatively thin. These plants are also uniformly chlorotic (yellowish color or lacking chlorophyll).
Diagnosing S deficiency requires proper plant tissue analyses. Gavlak et al. (1994) recommend a 2% (v/v) acetic acid extraction for New Mexico and other western states. Plant leaves can be tested for organic N and organic S content. A high ratio of organic N to organic S (70:1-80:1), as compared to healthy tissue (<50:1), is considered a diagnostic test since the S-containing amino acids are not formed in S-deficient plants. Another diagnostic tool for confirming S-deficient plants is an accumulation of nitrate-N. Plants with lower total protein and sugars in the leaf blades and stems can have a surplus of nitrate-N in the tissue (Ergle and Eaton, 1951).
Plant tissue levels for total S have different minimum levels for optimum production. Wheat, sunflower, and field beans have some of the highest requirements, whereas corn and soybeans have some of the lowest critical levels (Hitsuda et al., 2005) (Table 1).
Table 1. Critical Plant S Concentration for S Deficiency with 75% Relative Shoot Dry Weight Compared to Optimum Plant Growth (adopted from Hitsuda et al., 2005, and as referenced by Dick et al., 2008)
| Crop | Plant part and growth stage | Shoot S† concentration | |
|
Critical growth mg g-1 |
Optimum growth mg g-1 | ||
| Alfalfa (Medicago sativa L.) | Top 6 in. at early bud | 2.0 | 2.6-5.0 |
| Corn (Zea mays L.) | Ear leaf at silking | 1.0 | 2.1-5.0 |
| Corn | Shoot 30 days after emergence | 0.76 | 2.06 |
| Cotton (Gossypium hirsutum L.) | Youngest mature leaf blade at early flower | <2.0 | 2.0-2.5 |
| Cotton | Shoot 30 days after emergence | 1.07 | 4.1 |
| Field bean (Phaseolus vulgaris L.) | Shoot 30 days after emergence | 1.56 | 2.56 |
| Oat (Avena sativa L.) | Top leaves at boot stage | <1.5 | 2.1-4.0 |
| Sorghum (Sorghum bicolor (L.) Moench) | Shoot 30 days after emergence | 1.13 | 1.99 |
| Peanut (Arachis hypogaea L.) | Youngest mature leaf at preflower | <2.0 | 2.0-3.5 |
| Sunflower (Helianthus annuus L.) | Shoot 30 days after emergence | 1.56 | 3.12 |
| Wheat (Triticum aestivum L.) | Shoot 30 days after emergence | 1.43 | 2.33 |
| †Determined by spectrometer after wet washing dried tissue with nitric and perchloric acid. | |||
Plant tissue analysis interpretations should reflect these differences among crops. Mathot et al. (2009) have also recently proposed three diagnostic zones for total S and total N content for determining if grasses are S-deficient. Their guidelines include the effects of N nutrition and the dilution of the grass S content during growth. Grasses are S-deficient, in their proposed diagnostic tool, when the total S content (mg/g) falls on or below a line described by the equation 0.0665 x N - 0.2805 (total N is also presented in mg/g) (Figure 1). Validation of this tool is needed for New Mexico. Please share your results with your local county Extension agriculture agent.
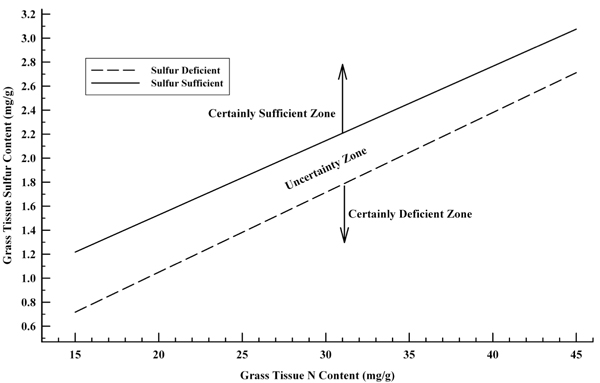
Figure 1. Boundary lines for identifying S sufficiency and S deficiency in grasses when given grass tissue total N and total S content in mg/g (adapted from Mathot et al., 2009).
The S content of plants is approximately the same as the P content (Mengel and Kirkby, 1982). However, S fertilization is not as critical as P since SO4-2 is much more mobile and not as strongly fixed to soil particles as P. Alfalfa, a major crop in New Mexico, has a relatively high S requirement. Approximately 5.3 lb of S are removed for every ton of harvested alfalfa (Chen et al., 2005). A ten ton yield goal for the Dona Ana County region of New Mexico would be expected to remove 50 to 55 lb/ac of S. Westermann (1975) reported that the critical biomass value for alfalfa is 0.15 to 0.20% S. Cotton lint can remove nearly 53 lb S/ac with a 3.2 bale per acre yield (Dick et al., 2008). The youngest, most fully developed leaf of cotton at early flowering should have between 0.20 and 0.25% S to be considered sufficient.
Sulfate Sources in New Mexico
Soil
Plants derive sulfate from soil solution SO4-2 and adsorbed inorganic SO4-2 (Kowalenko and Grimmett, 2008). Inorganic sulfate may be present in soil water, bound or adsorbed on soil surfaces, bound to gypsum (Nelson, 1982), or associated with calcium carbonate (Roberts and Bettany, 1985). Determining plant-available SO4-2, along with the plant-available N, in the soil can give the grower a "snapshot" of whether or not additional SO4-2 is needed for plant growth. Soils are generally considered sufficient in SO4-2 if the ratio of plant-available SO4-2 to plant-available N is less than 15:1.
There are a host of soil extracts that can be used to help determine plant-available SO4-2, including acetates, carbonates, chlorides, citrates, and oxalates. The preferred extraction method uses calcium phosphate (Beaton et al., 1968), which is used by the NMSU Soil Water Agricultural Testing laboratory. The phosphate ion is used to displace sulfate on soil adsorption sites. Many New Mexico soils, however, fix considerable amounts of phosphate, and labs must compensate by increasing the phosphate concentration in the extract. This "snapshot" does not account for sulfate that may become available from organic matter or other soil inputs or sources. It also does not account for what can happen to sulfate during the growing season as it cycles through the soil, plant, and air systems.
Sulfur cycling (Figure 2) in soils is a complex and dynamic process as outlined by Eriksen (2008). Inputs to soil solution SO4-2 include atmospheric S, animal manures, fertilizers, and decomposition of organic sources. Sulfate-S from the soil solution can be absorbed by the plant, adsorbed to soil particles, converted into other stable inorganic compounds, or leached beyond the plant root zone. Attention to irrigation water management, however, is important for SO4-2 management since SO4-2 is easily leached through many New Mexico soils, particularly sandy or coarse-textured soils. It is also recommended that soils be sampled below the plow layer since SO4-2 can be stored at lower depths in the soil profile due to leaching.
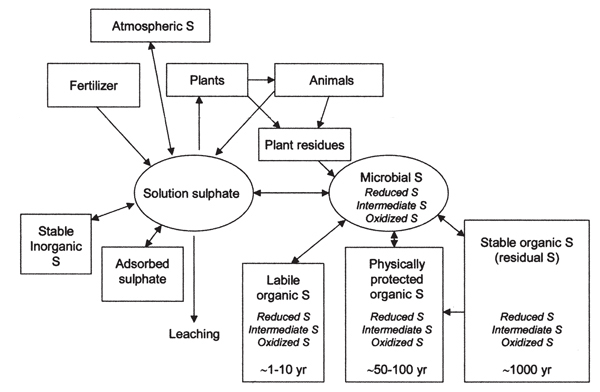
Figure 2. Conceptual model that demonstrates the soil sulfur cycle (from Eriksen, 2008; used with permission).
Irrigation Water
Irrigation water can be a major source of S for New Mexico fields. Table 2 illustrates the SO4-2-S variability from selected locations in New Mexico. An irrigation water sample with 4.5 mg/L SO4-2 would add approximately 1 lb SO4-2 per acre inch of applied water.
Table 2. Sulfate Concentration in Selected Waters Used for Irrigation
| Location | mg/L SO4-2 | lb/acre inch |
| Otero County | 1,921 | 427 |
| San Miguel County | 5 | 1.1 |
| Lea County | 767 | 170 |
| Doña Ana County | 1,440 | 320 |
| Eddy County | 1,100 | 244 |
Gypsum
Calcium sulfate (CaSO4.2H2O), or gypsum, is slightly soluble in water (~2 g/L = 8.3 lb per 1,000 gal; National Institute of Occupational Safety and Health, 2003) and can therefore provide some sulfate for plants. Many soils in New Mexico are gypsiferous (>2% calcium sulfate, approximately 76,134 lb gypsum per acre) and are usually not prone to S deficiency. A soil with at least 2% gypsum should have at least 107 lb of SO4-2 available for uptake under adequately watered conditions. Gypsum is used primarily to reclaim sodium (Na)-affected soils to provide the Ca needed to displace Na. A side benefit can be the SO4-2 for plant uptake. However, SO4-2 is a soluble anion that will contribute to soil salinity.
Elemental Sulfur
Oxidation of elemental sulfur (So) to plant-available SO4-2-S is mediated by soil microorganisms, primarily Thiobacillus spp. Oxidation of sulfur is influenced by its particle size, as well as the soil's temperature and moisture content. Warm, moist soil conditions are needed for optimal biological oxidation to occur, but oxidation will occur slowly in cool, moist soils. Additionally, the rate of So oxidation generally increases as the organic matter content increases. Several So sources have been used in New Mexico in an effort to affect soil pH, micronutrient availability, and SO4-2-S content. Lindemann et al. (1991) evaluated several S sources, including reagent-grade So, wettable S (90% So), Disper-Sul S (90% So), and flowable S (52% So), on calcareous soils. The small particle size (<425 µm) of the flowable S was more effective than the other sources of So when tested in the field, but only decreased pH by 0.1 to 0.2 units, which probably had no biological significance. However, the flowable form kept the pH at a lower level for more than 12 weeks after application, which was significantly longer than all other S sources. Wettable S increased soil salinity by 2 mmhos/cm, which could be harmful to salt-sensitive plants. The SO4-2 content increased by 1,000 ppm (mg/kg) as a result of using any of the tested S sources. This level of SO4-2 gradually decreased as the soil was irrigated and the SO4-2 leached from the root zone.
Organic Matter
The addition of organic matter can improve soil SO4-2-S content, depending on the composition and source of the organic matter (Cifuentes and Lindemann, 1993). When added to a clay soil, fresh cow manure (0.30% S) or bermudagrass clippings (0.22% S) resulted in 63.5% more soil SO4-2² than composted horse manure (0.10% S) during the first 8 weeks after application. The difference gradually decreased to just 9.4%, probably reflecting a loss of SO4-2 by leaching. Greater So oxidation in the presence of fresh cow manure or bermudagrass clippings was attributed to their higher N content and their ease of decomposition, which allowed for more decomposition of the S-containing carbon compounds. The higher S content and faster decomposition probably promoted a larger number of chemoheterotrophic S microbes than the more resistant composted horse manure (Cifuentes and Lindemann, 1993). Generally speaking, manures and plant material should have a relatively high N and S content to promote more plant-available SO4-2. It is difficult, however, to predict how much plant-available SO4-2 can come from added organic sources of S because of complicated dynamics in the soil environment (Eriksen, 2008; Scherer, 2001).
Other Sulfate Amendments
Many methods are available to increase soil organic matter, including green manures (turning cover crops into the soil), reduced tillage or no-tillage, and adding organic matter from farmyard manures.
Sulfate-containing fertilizers include, but are not limited to,
- ammonium sulfate (24% S),
- ammonium thiosulfate (26% S),
- ordinary superphosphate (11-12% S),
- magnesium sulfate (14% S),
- sulfate of potash magnesia (22% S), and
- potassium sulfate (18% S).
Sulfates and Groundwater Quality
Ground and surface water quality are regulated under the authority of chapter 6, part 2 of New Mexico's Title 20 environmental protection regulations (New Mexico Administrative Code, 2001). The standards for groundwater with less than 10,000 mg/L of total dissolved solids (TDS) limit the SO4-2² concentration for domestic water supplies to less than 600 mg/L unless the existing condition (natural state) exceeds that standard. Additionally, domestic water supplies must not exceed 1,000 mg TDS/L.
Sulfate and Cattle
The National Research Council (1996) gives the daily S requirement for cattle as 0.15% of diet dry matter, and the maximum tolerable concentrations of dietary S are estimated to be 0.40% of dry matter intake. Runyan et al. (2009) and Ellis (2008) offer guidelines for sulfate in drinking water for cattle (Table 3).
Table 3. Sulfate Levels in Drinking Water for Cattle
| Sulfate level (mg/L) |
Interpretation |
| <500 | Safe for drinking |
| 500-1,500 | Generally safe; trace mineral availability may be reduced; may decrease performance in confined cattle |
| 1,500-3,000 | Marginal; may be unsuitable for confined cattle during hot weather; performance may be reduced; sporadic cases of polioencephalomalacia (PEM) may occur |
| 3,000-4,000 | Unsuitable; decreased performance of grazing cattle may occur, and risk for PEM in confined cattle is increased |
| >4,000 | Dangerous; health problems expected and substantial reductions in cattle performance; secondary copper deficiency likely |
Best Management Practices
Sulfate plays a critical role in plant and animal health in New Mexico. Long-term success in growing plants includes knowing whether or not SO4-2 is needed. A sustainable approach to managing sulfate nutrition includes the following actions.
- Test the soil for plant-available SO4-2.
- Test the irrigation water as a viable source of SO4-2 for plants.
- Evaluate the plant tissue S content for critical levels at specific growth stages.
- Evaluate crop rotations that conserve or enhance SO4-2 nutrition, such as high residue crops that return SO4-2 sources to the soil or are not heavy SO4-2 users under SO4-2-limited conditions.
- Evaluate management practices that can build soil organic matter, which would include the responsible use of manures, green manures, and cover crops.
- Manage irrigation water to avoid excess leaching that could reduce soil SO4-2.
- Consider SO4-2 amendments if all the possible sources and plant requirements do not balance.
References
Beaton, J.D., G.R. Burns, and J. Platou. 1968. Determination of sulfur in soils and plant material [Technical Bulletin No. 14]. Washington, D.C.: The Sulfur Institute.
Chen, L., W. Dick, and S. Nelson. 2005. Flue gas desulfurization products as sulfur sources for alfalfa and soybean. Agronomy Journal, 97, 265-271.
Cifuentes, F.R., and W.C. Lindemann. 1993. Organic matter stimulation of elemental sulfur oxidation in a calcareous soil. Soil Science Society of America Journal, 57, 727-731.
Dick, W.A., D. Kost, and L. Chen. 2008. Availability of sulfur to crops from soil and other sources. In J. Jez (Ed.), Sulfur: A missing link between soils, crops, and nutrition [Agronomy Monograph 50], pp. 59-82. Madison, WI: American Society of Agronomy.
Ellis, R.W. 2008. Water quality issues for beef cattle.
Ergle, D.R., and F.M. Eaton. 1951. Sulfur nutrition of cotton. Plant Physiology, 26, 639-654.
Eriksen, J. 2008. Soil sulfur cycling in temperate agricultural systems. In J. Jez (Ed.), Sulfur: A missing link between soils, crops, and nutrition [Agronomy Monograph 50], pp. 25-44. Madison, WI: American Society of Agronomy.
Gavlak, R.G., D.A. Horneck, and R.O. Miller. 1994. Plant, soil and water reference methods for the western region [Western Regional Extension Publication 125]. Fairbanks: University of Alaska.
Hitsuda, K., M. Yamada, and D. Klepker. 2005. Sulfur requirement of eight crops at early stages of growth. Agronomy Journal, 97, 155-159.
Kowalenko, C.G., and M. Grimmett. 2008. Chemical characterization of soil sulfur. In M.R. Carter and E.G. Gregorich (Eds.), Soil sampling and methods of analysis, pp. 251-264. Pinawa, Manitoba: Canadian Society of Soil Science.
Lindemann, W.C., J.J. Aburto, W.M. Haffner, and A.A. Bono. 1991. Effect of sulfur source on sulfur oxidation. Soil Science Society of America Journal, 55, 85-90.
Mathot, M., L. Thélier-Huché, and R. Lambert. 2009. Sulphur and nitrogen content as sulphur deficiency indicator for grasses. European Journal of Agronomy, 30, 172-176.
McGrath, S.P., and F.J. Zhao. 1995. A risk assessment of sulfur deficiency in cereals using soil and atmospheric deposition data. Soil Use and Management, 11, 110-114.
Mengel, K., and E.A. Kirkby. 1982. Principles of plant nutrition. Bern, Switzerland: International Potash Institute.
National Institute of Occupational Safety and Health. 2003. NIOSH bibliography of communication and research products. Atlanta, GA: Author.
National Research Council. 1996. Nutrient requirements of beef cattle, seventh revised edition. Washington, D.C.: National Academy Press.
Nelson, R.E. 1982. Carbonate and gypsum. In A.L. Page, R.H. Miller, and D.R. Keeney (Eds.), Methods of soil analysis, part 2-Chemical and microbiological properties, 2nd ed., pp. 181-197. Madison, WI: American Society of Agronomy.
New Mexico Administrative Code. 2001. Ground and surface water protection. 20.6.2 NMAC.
Roberts, T.L., and J.R. Bettany. 1985. The influence of topography on the nature and distribution of soil sulfur across a narrow environmental gradient. Canadian Journal of Soil Science, 65, 419-434.
Runyan, C., J. Bader, and C. Mathis. 2009. Water quality for livestock and poultry [Guide M-112]. Las Cruces: New Mexico Cooperative Extension Service.
Scherer, H.W. 2001. Sulfur in crop production-Invited paper. European Journal of Agronomy, 14, 81-111.
Westermann, D.T. 1975. Indexes of sulfur deficiency in alfalfa. II. Plant analyses. Agronomy Journal, 67, 265-268.
To find more resources for your business, home, or family, visit the College of Agricultural, Consumer and Environmental Sciences on the World Wide Web at pubs.nmsu.edu.
Contents of publications may be freely reproduced for educational purposes. All other rights reserved. For permission to use publications for other purposes, contact pubs@nmsu.edu or the authors listed on the publication.
New Mexico State University is an equal opportunity/affirmative action employer and educator. NMSU and the U.S. Department of Agriculture cooperating.
Printed and electronically distributed March 2010, Las Cruces, NM.
