CR 656
Robert Flynn and April Ulery
College of Agricultural, Consumer and Environmental Sciences, New Mexico State University
Authors: Respectively, Associate Professor and Extension Agronomist, Department of Extension Plant Sciences; and Professor and Soil Scientist, Department of Plant and Environmental Sciences, both of New Mexico State University. (Print Friendly PDF)
Overview
Most of us are familiar with table salt, Epsom salt, and sea salt, but there are many kinds of salts, including several fertilizers such as ammonium sulfate, potassium chloride, and potassium sulfate. Technically, salts are ionic compounds formed by the neutralization of an acid and a base. Salts are necessary for providing many of the minerals that both plants and humans need in order to be healthy. Too much salt is unhealthy for humans as well as plants. High salt in the plant root zone interferes with the uptake of water and can cause death. It does not matter to the plant what kind of salt it is. Thankfully, plants have different levels of tolerance to salts found in the soil or in irrigation water. Since salts can conduct electricity when dissolved in water, we can measure the total "saltiness" of a soil by using a water extract and measuring how well the water conducts electricity. We can combine this measure of saltiness with knowledge of how plants respond to the salt to improve productivity using optimum soil management and plant selection.
Soil Electrical Conductivity
Electricity is used to measure the salt content of water-based solutions. If you have worked with electricity, you are probably familiar with the ohm meter, which measures electrical resistance. Conductance is the opposite of resistance and is measured in units of mho (ohm, unit of resistance, spelled backward). Conductance is measured over a known distance, and in soil extracts we report conductance as millimhos per centimeter (mmhos/cm). The conductivity of a solution increases as the amount of salt increases. Pure water does not conduct electricity, but if the tiniest bit of salt is present, electricity is conducted through the solution. This is referred to as electrical conductivity (EC). The scientific community uses the units of deciSiemens per meter (dS/m), which are the same magnitude as mmhos/cm. EC measurements give us an idea of how salty the water is but do not tell us what kinds of salts are present. Some salts conduct electricity better than others, just as some metals conduct electricity better than others. Table 1 presents some observations made after mixing selected fertilizers into water and measuring the conductivity of each solution.
Table 1. Salinity of Common Fertilizers Using 1 Tablespoon of Product Per Quart of Distilled Water (unless otherwise noted)
| Source |
Electrical Conductivity
(mmhos/cm)
|
| Distilled water |
0
|
| Tap water |
Variable
|
| Urea (46-0-0) |
4
|
| 15-30-15 (1 teaspoon/gal) |
10
|
| Mono-ammonium phosphate (11-52-0) |
81
|
| Ammonium nitrate (32-0-0) |
191
|
| Muriate of potash (0-0-60) |
210
|
| Ammonium sulfate (21-0-0) |
230
|
Sampling Soil to Assess Salinity
It is critical to collect a representative sample for the area to be managed to determine what management practices are needed. Grid sampling can help identify areas that are more salty than others. If your field or garden has distinct areas that are higher or lower in elevation, or managed differently, then each area must be sampled individually.
Plants are most sensitive to soil salinity when the seed is germinating. A soil sample should reflect the conditions around the seed, so sampling no deeper than 2 inches is suggested if you are concerned about germination and plant establishment. However, for purposes of cost and assessing the potential root zone for nutrients, sampling from the surface to 8 or 12 inches is acceptable. The soil should be sampled at least 6 to 8 weeks prior to planting.
Lab Methods – Saturated Paste
The standard practice for determining soil salinity or "saltiness" is to saturate the soil with distilled pure water (i.e., fill all of the pores in between soil particles with water), and then extract the water from the saturated soil paste after 24 hours. The EC determined from this procedure is abbreviated ECe. Some labs will mix one part water with one part soil by volume or weight to test for soil salts. This method takes less time than the saturated paste method. However, the saturated paste method is best for evaluating crop response to soil salinity since most reference tables are based on this procedure. Other methods, or different ratios of soil to water, are suitable for relative comparisons. The main reason why the saturated paste method works so well has to do with the solubility of salts in soil. Too much water could dilute the effects of salt on plant/water relationships seen in the saturated soil environment. The saturated paste is a compromise between having enough liquid to extract for analysis while still reflecting the soil conditions that a plant would experience after being watered. A comparison of several soils from around New Mexico suggests that the higher the salinity level, the greater the difference between the two methods (Figure 1).
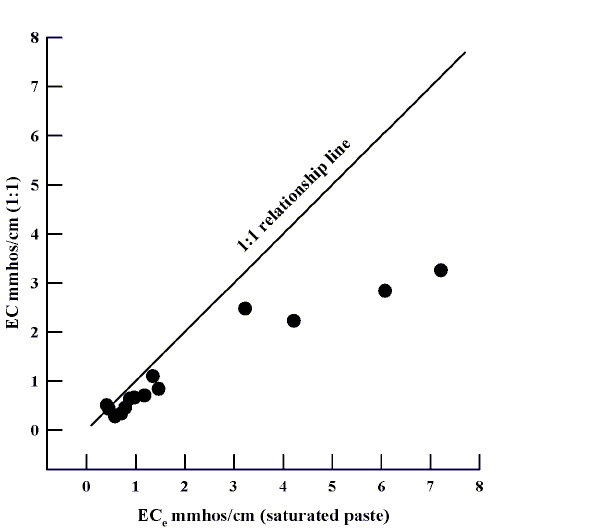
Figure 1. Soil salinity measured by 1:1 water:soil and by saturated paste methods. Note that the methods do not give the same result as salinity increases in a soil.
Interpretation of Electrical Conductivity
Electrical conductivity values do not mean anything until they are interpreted or correlated to plant responses such as growth or yield. However, the higher the EC, the fewer the types of plants that will grow well. Most ornamental and crop plants do not thrive in saline soils with high EC. In general, a soil is saline if the conductivity of the saturated paste extract is greater than 4 mmhos/cm or dS/m. Saline soils do not mean that an excess of sodium is present. Saline soils have an exchangeable sodium percentage (ESP) less than 15%. However, sodium reclamation procedures should begin when ESP values are greater than 6%. Excess sodium causes its own set of problems and warrants special attention. The pH of a saline soil is also usually less than 8.2.
Procedure for Determining Salinity Thresholds of Plants
Soil salinity disrupts the water balance in plants. Plants generally respond to soil salinity with reduced growth and transpiration rates. However, not all plants respond the same to soil salinity because of myriad adaptations and biological processes that can improve plant tolerance to soil salinity. Plants are evaluated for their tolerance by growing them in controlled experiments where the soil salinity is varied in different containers or plots while other growing conditions are the same. The plants are all watered, fertilized, and grown the same way for a pre-determined amount of time. Plants are then harvested for dry matter or yield and plotted against the soil salinity as measured by EC (Figure 2). The salinity level at which plants are first affected by salinity is referred to as the "threshold." The threshold varies with crop tolerance and external environmental factors that affect the plant's need for water. The rate at which the yield decreases as salinity increases is the "slope." The threshold and slope reflect how sensitive the plant is to salinity. Armed with the threshold and slope of a given plant, as well as the soil salinity from a soil test, we can predict how well a plant will grow in that soil. Rhoades et al. (1992) present the following equation to estimate relative yield. (A relative yield of 100% implies no salinity effect on plant yield or expected performance. A relative yield of 90% implies a 10% reduction in plant performance due to salinity, or 90% of expected plant productivity without salinity.)
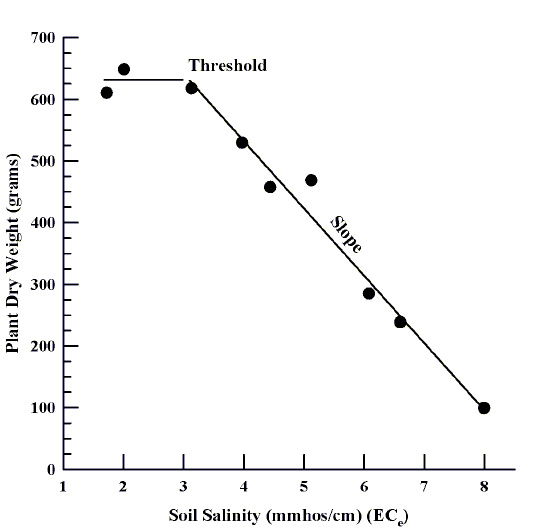
Figure 2. Chile plant response to increasing soil salinity.
(Equation 1)
100 – ((soil test ECe – threshold) × slope) = relative yield
Example:
Chosen plant: Scallop squash (See Table 2)
Threshold of chosen plant: 3.2 mmhos/cm
Slope of chosen plant: 16
Soil test value for salinity from client 1
(Soil 1): 4.5 mmhos/cm
Soil test value for salinity from client 2
(Soil 2): 2.3 mmhos/cm
Soil 1: 100 – ((4.5 – 3.2) × 16) = (1.3 × 16) = 79.2% relative yield
Soil 2: 100 – ((2.3 – 3.2) × 16) = >100%, or no negative effect of salinity on plant performance
It can also be said of Soil 1 that there is a 20.8% reduction in plant performance, while there is no reduction in plant performance for Soil 2.
Vegetable responses to salinity are given in Table 2, grasses in Table 3, woody species in Table 4, and forage and field crops in Table 5. To use the tables, find the plant of interest and move to the right. The third column is the conductivity value at which the plant is first affected by soil salts (threshold). The higher this number is, the more tolerant the plant is to salt. The next column (column 4) is the slope, which tells you how much reduction in yield or performance can be expected to occur for every unit of increased salinity above the plant's threshold EC. The last column rates the plant as sensitive (S), moderately sensitive (MS), moderately tolerant (MT), or tolerant (T) to soil salt levels. Ratings are also used as estimates when thresholds and slopes are not available from published values.
Table 2. Tolerance of Selected Vegetables to Soil Salinitya (Under saline conditions, plant only those plants that are rated tolerant [MT or T].)
|
Common Name
|
Botanical Nameb
|
Thresholdc (mmhos/cm)
|
Slope (%/mmhos/cm)
|
Ratingd
|
| Artichoke | Cynara scolymus |
—
|
—
|
MT*
|
| Asparagus | Asparagus officinalis |
4.1
|
2
|
T
|
| Bean | Phaseolus vulgaris |
1
|
19
|
S
|
| Bean, mung | Vigna radiata |
1.8
|
20.7
|
S
|
| Beet, red | Beta vulgaris |
4
|
9
|
MT
|
| Broccoli | Brassica oleracea italica |
2.8
|
9.2
|
MS
|
| Brussels sprouts | B. oleracea gemmifera |
—
|
—
|
MS*
|
| Cabbage | B. oleracea capitata |
1.8
|
9.7
|
MS
|
| Carrot | Daucus carota |
1
|
14
|
S
|
| Cauliflower | Brassica oleracea botrytis |
—
|
—
|
MS*
|
| Celery | Apium graveolens |
1.8
|
6.2
|
MS
|
| Corn, sweet | Zea mays |
1.7
|
12
|
MS
|
| Cucumber | Cucumis sativus |
2.5
|
13
|
MS
|
| Eggplant | Solanum melongena |
1.1
|
6.9
|
MS
|
| Kale | Brassica oleracea acephala |
—
|
—
|
MS*
|
| Kohlrabi | B. oleracea gongylodes |
—
|
—
|
MS*
|
| Lettuce | Lactuca sativa |
1.3
|
13
|
MS
|
| Muskmelon | Cucumis melo |
—
|
—
|
MS
|
| Okra | Abelmoschus esculentus |
—
|
—
|
S
|
| Onion | Allium cepa |
1.2
|
16
|
S
|
| Parsnip | Pastinaca sativa |
—
|
—
|
S*
|
| Pea | Pisum sativum |
—
|
—
|
S
|
| Peppere | Capsicum annuum |
1.5
|
14
|
MS
|
| Potato | Solanum tuberosum |
1.7
|
12
|
MS
|
| Pumpkin | Cucurbita pepo |
—
|
—
|
MS*
|
| Radish | Raphanus sativus |
1.2
|
13
|
MS
|
| Spinach | Spinacia oleracea |
2
|
7.6
|
MS
|
| Squash, scallop | Cucurbita pepo melopepo |
3.2
|
16
|
MS
|
| Squash, zucchini | C. pepo melopepo |
4.7
|
9.4
|
MT
|
| Strawberry | Fragaria spp. |
1
|
33
|
S
|
| Sweet potato | Ipomoea batatas |
1.5
|
11
|
MS
|
| Tomato | Lycopersicon lycopersicum |
2.5
|
9.9
|
MS
|
| Tomato, cherry | L. esculentum var. cerasiforme |
1.7
|
9.1
|
MS
|
| Turnip | Brassica rapa |
0.9
|
9
|
MS
|
| Watermelon | Citrullus lanatus |
—
|
—
|
MS*
|
| aThese data serve only as a guideline to relative tolerances among crops. Absolute tolerances vary depending on climate, soil conditions, and cultural practices. bBotanical and common names follow the convention of Hortus Third (Liberty Hyde Bailey Hortorium Staff, 1976) where possible. cIn soils containing >2% gypsum, plants will tolerate ECe about 2 mmhos/cm higher than indicated. dS = sensitive, MS = moderately sensitive, MT = moderately tolerant, T = tolerant. Ratings with an * are estimates (Maas, 1986).eDifferences exist among varieties. |
||||
Table 3. Tolerance of Selected Grass Species to Soil Salinitya (Mass, 1986) (Common names denoted with † use the National Plants Database to estimate salinity tolerance. Under saline conditions, plant only those plants that are rated tolerant [MT or T].)
Lovegrassf|
Common Name
|
Botanical Nameb
|
Thresholdc (mmhos/cm)
|
Slope (%/mmhos/cm)
|
Ratingd
|
| Alkaligrass, Nuttall's |
Puccinellia airoides
|
—
|
—
|
T*
|
| Bentgrass |
Agrostis stolonifera palustris
|
—
|
—
|
MS
|
| Bermudagrasse |
Cynodon dactylon
|
6.9
|
6.4
|
T
|
| Bluegrass, Kentucky |
Poa pratensis
|
—
|
—
|
S
|
| Bluestem, big† |
Andropogon gerardii
|
4.1-8.0
|
—
|
MT*
|
| Bluestem, little† |
Schizachyrium scoparium
|
0-2
|
—
|
S
|
| Canarygrass, reed |
Phalaris arundinacea
|
—
|
—
|
MT
|
| Dallisgrass |
Paspalum dilatatum
|
—
|
—
|
MS*
|
| Fescue, tall |
Festuca elatior
|
3.9
|
5.3
|
MT
|
| Fescue, meadow |
Festuca pratensis
|
—
|
—
|
MT*
|
| Foxtail, meadow |
Alopecurus pratensis
|
1.5
|
9.6
|
MS
|
| Grama, blue |
Bouteloua gracilis
|
—
|
—
|
MS*
|
| Grama, black† |
Bouteloua eriopoda
|
2.1-4.0
|
—
|
MS*
|
| Grama, sideoats† |
Bouteloua curtipendula
|
2.1-4.0
|
—
|
MS*
|
| Indiangrass† |
Sorghastrum nutans
|
4-8
|
—
|
MT
|
| Orchardgrass |
Dactylis glomerata
|
1.5
|
6.2
|
MS
|
| Ryegrass, perennial |
Lolium perenne
|
5.6
|
7.6
|
MT
|
| Sacaton, alkali |
Sporobolus airoides
|
—
|
—
|
T*
|
| Sacaton, big† |
Sporobolus wrightii
|
2.1-4.0
|
—
|
MS*
|
| Saltgrass, desert |
Distichlis stricta
|
—
|
—
|
T*
|
| Switchgrass† |
Panicum virgatum
|
4.1-8.0
|
—
|
MT*
|
| Wheatgrass, tall |
Thinopyrum ponticum
|
7.5
|
4.2
|
T
|
| Wheatgrass, slender† |
Elymus trachycaulus
|
>8
|
—
|
T
|
| Wheatgrass, intermediate† |
Thinopyrum intermedium
|
4.1-8.0
|
—
|
MT*
|
| Wheatgrass, western† |
Pascopyrum smithii
|
>8
|
—
|
T
|
| Wheatgrass, crested† |
Agropyron cristatum
|
4.1-8.0
|
—
|
MT
|
| Wheatgrass, desert† |
Agropyron desertorum
|
4.1-8.0
|
—
|
MT*
|
| aThese data serve only as a guide to relative tolerances among grasses. Absolute tolerances vary depending on climate, soil conditions, and cultural practices. bBotanical and common names follow the convention of Hortus Third (Liberty Hyde Bailey Hortorium Staff, 1976) where possible. cIn soils containing >2% gypsum, plants will tolerate ECe about 2 mmhos/cm higher than indicated. dS = sensitive, MS = moderately sensitive, MT = moderately tolerant, T = tolerant. Ratings with an * are estimates. eAverage of several cultivars. 'Suwannee' and 'Coastal' cultivars are about 20% more tolerant, and common and 'Greenfield' are about 20% less tolerant than the average. fAverage for 'Boer', 'Wilman', sand, and weeping cultivars. 'Lehmann' seems about 50% more tolerant. |
||||
Table 4. Tolerance Rating from the National Plants Database (https://plants.usda.gov) for Selected Woody Speciesa Found in New Mexico (Other characteristics of most of these woody species can be found in NMSU Guide H-328, Selecting Ornamental Trees for New Mexico, available at https://pubs.nmsu.edu/_h/H-328.pdf. Common names and characteristics denoted with † are from Maas [1986].)
|
Common Name
|
Botanical Name
|
Thresholdb (mmhos/cm)
|
Slope (%/mmhos/cm)
|
Ratingc
|
| Apple† | Malus sylvestris |
—
|
—
|
S
|
| Apricot† | Prunus armeniaca |
1.6
|
24
|
S
|
| Ash, European mountain | Sorbus aucuparia |
0-2
|
—
|
S
|
| Ash, green | Fraxinus pennsylvanica |
2.1-4.0
|
—
|
MS
|
| Ash, singleleaf | Fraxinus anomala |
2.1-4.0
|
—
|
MS
|
| Ash, velvet | Fraxinus velutina |
0-2
|
—
|
S
|
| Aspen, quaking | Populus tremuloides |
4.1-8.0
|
—
|
MT
|
| Basswood, American (Linden) | Tilia americana |
0-2
|
—
|
S
|
| Birch, water | Betula occidentalis |
0-2
|
—
|
S
|
| Blackberry† | Rubus spp. |
1.5
|
22
|
S
|
| Buckthorn, redberry | Rhamnus crocea |
—
|
—
|
S
|
| Catalpa, northern | Catalpa speciosa |
2.1-4.0
|
—
|
MS
|
| Cherry, sweet† | Prunus avium |
0-2
|
—
|
S*
|
| Cherry, sand† | Prunus besseyi |
0-2
|
—
|
S*
|
| Chinaberry (Texas umbrella) | Melia azedarach |
4.1-8.0
|
—
|
MT
|
| Chokecherry | Prunus virginiana |
4.1-8.0
|
—
|
MT
|
| Chokecherry, western | Prunus virginiana L. var. demissa |
4.1-8.0
|
—
|
MT
|
| Coffeetree, Kentucky | Gymnocladus dioica |
4.1-8.0
|
—
|
MT
|
| Cottonwood, Fremont (valley) | Populus fremontii |
2.1-4.0
|
—
|
MS
|
| Currant† | Ribes spp. |
—
|
—
|
S*
|
| Elm, Chinese | Ulmus parvifolia |
0-2
|
—
|
S
|
| Fig† | Ficus carica |
—
|
—
|
MT*
|
| Grape† | Vitis spp. |
1.5
|
9.6
|
MS
|
| Guayule† | Parthenium argentatum |
15.0
|
13
|
T
|
| Hackberry, common | Celtis occidentalis |
2.1-4.0
|
—
|
MS
|
| Hackberry, netleaf (western) | Celtis reticulata |
2.1-4.0
|
—
|
MS
|
| Hawthorn, cerro | Crataegus erythropoda |
4.1-8.0
|
—
|
MT
|
| Hawthorn, Washington | Crataegus phaenopyrum |
0-2
|
—
|
S
|
| Honeylocust, thornless | Gleditsia triacanthos |
4.1-8.0
|
—
|
MT
|
| Hoptree, common | Ptelea trifoliata |
4.1-8.0
|
—
|
MT
|
| Hornbeam, American | Carpinus caroliniana |
0-2
|
—
|
S
|
| Jojoba† | Simmondsia chinensis |
—
|
—
|
T
|
| Jujube† | Ziziphus jujuba |
—
|
—
|
MT*
|
| Linden, littleleaf | Tilia cordata |
0-2
|
—
|
S
|
| Locust, New Mexico | Robinia neomexicana |
2.1-4.0
|
—
|
MS
|
| Loquat† | Eriobotrya japonica |
—
|
—
|
S*
|
| Madrone, Arizona | Arbutus arizonica |
2.1-4.0
|
—
|
MS
|
| Maidenhair tree | Ginkgo biloba |
2.1-4.0
|
—
|
MS
|
| Maple, bigtooth | Acer grandidentatum |
—
|
—
|
S
|
| Maple, silver | Acer saccharinum |
2.1-4.0
|
—
|
MS
|
| Maple, sugar | Acer saccharum |
0-2
|
—
|
S
|
| Mescal bean | Sophora secundiflora |
>8
|
—
|
T
|
| Mesquite, honey | Prosopis glandulosa |
2.1-4.0
|
—
|
MS
|
| Oak, bur | Quercus macrocarpa |
2.1-4.0
|
—
|
MS
|
| Oak, chinquapin | Quercus muehlenbergii |
0-2
|
—
|
S
|
| Oak, Gambel | Quercus gambelii |
0-2
|
—
|
S
|
| Oak, live (southern) | Quercus virginiana |
2.1-4.0
|
—
|
MS
|
| Oak, Shumard | Quercus shumardii |
0-2
|
—
|
S
|
| Oak, valley (California white) | Quercus lobata |
0-2
|
—
|
S
|
| Passion fruit† | Passiflora edulis |
—
|
—
|
S*
|
| Peach† | Prunus persica |
1.7
|
21
|
S
|
| Pear, Callery (Bradford) | Pyrus calleryana |
2.1-4.0
|
—
|
MS
|
| Pecan | Carya illinoinensis |
2-3d
|
—
|
S
|
| Persimmon† | Diospyros virginiana |
—
|
—
|
S*
|
| Planetree, London | Platanus hybrida |
0-2
|
—
|
S
|
| Plum; prune† | Prunus domestica |
1.5
|
18
|
S
|
| Pomegranate† | Punica granatum |
—
|
—
|
MT*
|
| Raintree, golden | Koelreuteria paniculata |
2.1-4.0
|
—
|
MS
|
| Raspberry† | Rubus idaeus |
—
|
—
|
S
|
| Redbud, eastern | Cercis canadensis var. canadensis |
0-2
|
—
|
S
|
| Redbud, Texas | Cercis canadensis var. texensis |
2.1-4.0
|
—
|
MS
|
| Silk tree | Albizia julibrissin |
2.1-4.0
|
—
|
MS
|
| Soapberry, western | Sapindus saponaria L. var. drummondii |
0-2
|
—
|
S
|
| Stretchberry | Forestiera pubescens |
4.1-8.0
|
—
|
MT
|
| Sweetgum | Liquidambar styraciflua |
0-2
|
—
|
S
|
| Sycamore, American | Platanus occidentalis |
0-2
|
—
|
S
|
| Sycamore, Arizona | Platanus wrightii |
0-2
|
—
|
S
|
| Tuliptree (tulip poplar) | Liriodendron tulipifera |
2.1-4.0
|
—
|
MS
|
| Walnut, Arizona | Juglans major | 0-2 |
—
|
S
|
| Walnut, black | Juglans nigra |
0-2
|
—
|
S
|
| Walnut, English | Juglans regia |
0-2
|
—
|
S
|
| Willow, desert | Chilopsis linearis |
2.1-4.0
|
—
|
MS
|
| aThese data are applicable with rootstocks that do not accumulate sodium (Na+) or chloride (Cl-) rapidly, or when they do not predominate in soil. bIn soils containing >2% gypsum, plants will tolerate ECe about 2 mmhos/cm higher than indicated. When a range for threshold is given, please refer to the National Plants Database for more information unless otherwise indicated cS = sensitive, MS = moderately sensitive, MT = moderately tolerant, T = tolerant. Ratings with an * are estimates dMiyamoto, S., G.R. Gobran, and K. Piela. 1985. Salt effects on seedling growth and ion uptake of three pecan rootstock cultivars. Agronomy Journal, 77, 383-388. |
||||
Table 5. Salt Tolerance of Selected Herbaceous Crops or Cover Cropsa Found in New Mexico
|
Common Name
|
Botanical Nameb
|
Thresholdc (mmhos/cm)
|
Slope (%/mmhos/cm)
|
Ratingd
|
| Alfalfae | Medicago sativa |
2
|
7.3
|
MS
|
| Barleyf | Hordeum vulgare |
8
|
5
|
T
|
| Cotton | Gossypium hirsutum |
7.7
|
5.2
|
T
|
| Brome, mountain | Bromus marginatus |
—
|
—
|
MT*
|
| Brome, smooth | Bromus inermis |
—
|
—
|
MS
|
| Clover, red | Trifolium repens |
1.5
|
12
|
MS
|
| Clover, strawberry | Trifolium fragiferum |
1.5
|
12
|
MS
|
| Clover, white | Trifolium repens |
—
|
—
|
MS*
|
| Maize (Corn) | Zea mays |
1.7
|
12
|
MS
|
| Milkvetch, cicer | Astragalus cicer |
—
|
—
|
MS*
|
| Oats | Avena sativa |
—
|
—
|
MT*
|
| Peanut | Arachis hypogaea |
3.2
|
29
|
MS
|
| Safflower | Carthamus tinctorius |
—
|
—
|
MT
|
| Sorghum | Sorghum bicolor |
6.8
|
16
|
MT
|
| Sudangrass | Sorghum sudanense |
2.8
|
4.3
|
MT
|
| Sugarbeet | Beta vulgaris |
7
|
5.9
|
T
|
| Sunflower | Helianthus annuus |
—
|
—
|
MS*
|
| Sweetclover, annual yellow | Melilotus indicus |
—
|
—
|
MS
|
| Sweetclover, yellow | Melilotus officinalis |
—
|
—
|
T
|
| Timothy | Phleum pratense |
—
|
—
|
MS
|
| Triticale | ×Triticosecale rimpaui |
6.1
|
2.5
|
T
|
| Vetch, common | Vicia angustifolia |
3
|
11
|
MS
|
| Wheat | Triticum aestivum |
8.6
|
3
|
T
|
|
aThese data serve only as a guide to relative tolerances among crops. Absolute tolerances vary depending on cultivar, climate, soil conditions, and cultural practices. |
||||
Sodic Soils
Sodic soils are identified by testing for exchangeable calcium, magnesium, and sodium. A high amount of exchangeable sodium relative to calcium and magnesium coupled with high pH causes soil crumbs to disperse or break apart. Soil particles that are dispersed are much smaller than well-aggregated or clumped soil and cause the destruction of soil structure. The soil surface usually forms a crust, and pore spaces become clogged with tiny, dispersed soil particles that prevent water movement into and through the soil. Such soils are called "sodic" and are high in exchangeable sodium as compared to calcium and magnesium. The sodium adsorption ratio (SAR) is above 13 and the exchangeable sodium percentage (ESP) is above 15%. The EC is less than 4 mmhos/cm and often less than 2 mmhos/cm, so while the total amount of salts may not be that high, the proportion of sodium to other salts is what makes a soil sodic or not. The soil pH is often greater than 8.5. The physical condition of the soil may exhibit crusting and dispersion at the surface, but this can be affected by soil texture, organic matter, and the EC of the irrigation water. Sodic soils often have a black color due to dispersion of dark organic matter and a greasy or oily looking surface with little or no vegetative growth. These soils have been called "black alkali" or "slick spots."
Soils that have high salinity and high exchangeable sodium are referred to as "saline-sodic." The soil pH can be above or below 8.5, salinity is greater than 4 mmhos/cm, and the SAR is greater than 13. These soils may have good water infiltration, but must be managed carefully. Figure 3 illustrates that soils can be non-saline/non-sodic, saline/non-sodic, non-saline/sodic, or saline-sodic.
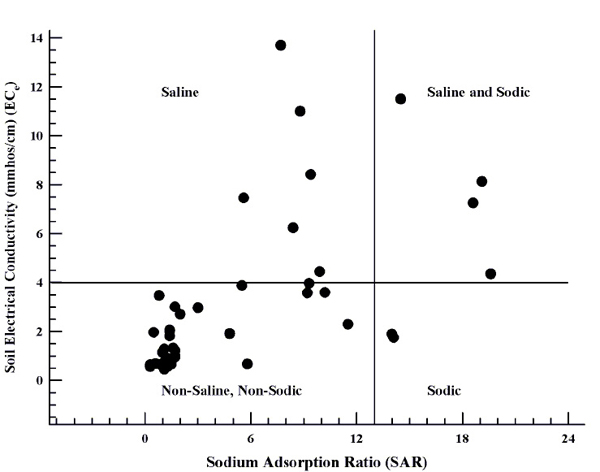
Figure 3. Salinity assessment of selected soils submitted to NMSU illustrating that soils can be non-saline/non-sodic, saline/non-sodic, non-saline/sodic, and saline-sodic.
Managing Salt-Affected Soil
Salt-affected soil problems do not develop overnight, nor are they solved quickly. It can take years for salt to accumulate to levels that reduce crop growth or water infiltration. Reclamation can take just as long. Before undertaking a reclamation program, develop a plan with a knowledgeable consultant. Understanding the implications and costs of a plan is important. In some situations it is not economical or even possible to reclaim a salt-affected soil.
Managing saline and sodic soils includes soil testing, irrigation water testing, water management, on-site drainage evaluation, and crop selection. Chemical amendments are not usually needed for saline soils. Amendments become necessary if sodium is excessively high in a soil test. If samples come back high in salinity, then excess water must be applied to move the salts away from the root zone, provided there is good drainage and no barriers to water movement through the soil. Signs of poor drainage include water ponding on the soil surface, slow infiltration, or soil that stays wet for a prolonged period of time. The leaching requirement can be calculated with the salinity of the irrigation water and the salinity of the soil.
Salt-affected soils are reclaimed by leaching salts beyond the root zone of the crop. It is impossible to lower the soil salt levels more than 1.5 times the salinity of the irrigation water (e.g., if your irrigation water has an EC of 2 mmhos/cm, then your soil EC will be about 1.5 × 2 = 3 mmhos/cm, provided you leach the soil with enough water). The greater the salinity of the irrigation water, the greater the quantity of water required to leach the salts to below the root zone. As a general rule, soil salinity is reduced by half for every 6 inches of good-quality water that moves through the soil. Good-quality water has an EC lower than the target EC for soil. It is critical to understand that reclamation of salty ground requires excess water. Six inches of water is approximately 163 gallons of water per 1,000 square feet. Monitoring soil EC following each leaching event will help you make adjustments to irrigation practices to lower soil salinity. Familiarity with the soil water holding capacity
Managing Sodic Soils
The same water management principles used in managing saline soils apply to sodic soils. Land with a high water table can be reclaimed if the water table can be lowered. Tile drains or altering the topography of the area can help drainage. Good irrigation water quality in this case means a source that is not very low in soluble salts. Low salinity water (<2 mmhos/cm) can make the problem worse for reducing sodium levels in sodic soils. Tillage is also often needed to physically break apart sodium-rich layers and mix in amendments. The addition of slowly decomposing organic matter such as straw, corn stalks, sawdust, or wood chips can help improve structure when combined with other amendments and reclamation practices.
The goal for the improvement of a sodic soil is to improve water infiltration. For a sodium-affected soil that is not saline, this means increasing the EC of the soil to more than 4 mmhos/cm or reducing the ESP. The ESP required to improve water infiltration depends on soil texture and irrigation method. Soils with high amounts of sand usually can tolerate higher ESPs (up to 12%) and still retain good water infiltration and percolation properties. Soils that are sprinkler irrigated typically require a lower ESP for good water infiltration compared to soils irrigated with surface systems.
Calcium must be added in order to lower the ESP. Calcium (a divalent ion) easily displaces sodium (a monovalent ion) on clay surfaces in the soil. Magnesium is also a divalent ion but is about half as effective as calcium at displacing sodium. High soil magnesium levels can also cause soils to disperse much like sodium does. Gypsum (calcium sulfate) is the most common amendment used to reclaim sodic soil. The gypsum requirement (GR) is an amount of gypsum required to lower the ESP to a given depth. General rates are given in Table 6 or can be calculated by using the following procedure.
Table 6. Rates of Gypsum and Elemental Sulfur as Amendments to Reclaim Sodic Soils to Two Different Soil Depths
| Exchangeable sodium to be replaced by calcium |
Gypsum
|
Elemental S
|
||||||
|
12 inches
|
6 inches
|
12 inches
|
6 inches
|
|||||
| meq/100 g soil |
ton/ac
|
lb/1,000 ft2
|
ton/ac
|
lb/1,000 ft2
|
ton/ac
|
lb/1,000 ft2
|
ton/ac
|
lb/1,000 ft2
|
|
1
|
1.7
|
80
|
0.9
|
40
|
0.35
|
16
|
0.17
|
8
|
|
2
|
3.5
|
159
|
1.7
|
80
|
0.69
|
32
|
0.35
|
16
|
|
4
|
6.9
|
318
|
3.5
|
159
|
1.39
|
64
|
0.69
|
32
|
|
8
|
13.9
|
636
|
6.9
|
318
|
2.77
|
127
|
1.39
|
64
|
|
16
|
27.7
|
1,273
|
13.9
|
636
|
5.54
|
255
|
2.77
|
127
|
Gypsum Rate Calculations
Theoretical gypsum application rates (Equation 2) can be calculated using soil test values for ESP and the cation exchange capacity (CEC), as well as actual or estimated bulk density and depth of soil to treat (Kamphorst and Bolt, 1978). However, we can simplify the equation by taking care of all the conversion factors needed to get to tons per acre and using an average bulk density of 1.48 g/cm3 and 1 foot of soil for the depth to treat. This works out to be 1.7 tons gypsum/ac/meq of Na+. The CEC can be estimated from soil texture, but it is best to have the soil testing laboratory determine what the soil CEC is. The ESP must also be determined by the soil testing lab and cannot be estimated. Hand calculations can be made using the following steps:
Given
Initial soil ESP = 15%
Soil CEC = 24 meq/100 g
Desired ESP = 6%
Purity of gypsum source = 90%
Determine amount of gypsum (100% pure)
How much sodium needs to be replaced?
1) initial soil ESP – desired ESP = 15%-6% = 9%
2) CEC × 9% = 24 × 0.09 = 2.16 meq Na+
3) tons gypsum per acre = 2.16 × 1.7 = 3.7 ton/ac
4) tons 90% pure gypsum per acre = 3.7 / 0.90 = 4.1 ton/ac
(Equation 2)
Gypsum rate = (current ESP – desired ESP) × CEC† × bulk density (g/cm3) × depth (cm) × 3.84
†Units for CEC should be meq/100 g soil or cmolc/kg soil.
Gypsum is used because it is calcium-rich (~23% by weight), is somewhat soluble in water at high pH, and does not contain elements or compounds that might interfere with reclamation. Gypsum applications can be made prior to reaching excessive levels of sodium to help prevent a problem before it gets worse. If soil ESP levels are at 6% or above, recommendations for gypsum are usually made. The sulfate in gypsum is not likely to be a problem for crops even though it is applied in quantities greatly in excess of plant needs.
Calcium nitrate or calcium chloride minerals can be used to reclaim sodic soils, but they generally are more costly and are likely to produce other negative effects on plant growth or the environment. Nitrate is considered a groundwater contaminant and is not a good choice. Limestone is another commonly available mineral that contains calcium and is naturally present in many New Mexico soils. However, it is not used for reclaiming sodic soils because it is not soluble at the high pH levels common in New Mexico soils and will increase the pH of soil that it is applied to. Theoretically, limestone as an amendment could be acidified to destroy the mineral bonds and release calcium, but this solution is both impractical and expensive.
Elemental sulfur (S) can be used for reclaiming sodic soil. Use elemental S only if free lime already exists in the soil. The amount of free lime in the soil can be determined at most soil testing labs, including NMSU's Soil, Water, and Agricultural Testing Laboratory (SWAT). The addition of S does not directly add calcium to the soil. However, elemental S oxidizes to form sulfuric acid in the presence of specific bacteria, which dissolves lime (calcium carbonate, CaCO3), often present in New Mexico soils. The dissolution of indigenous lime provides the calcium necessary to reclaim a sodic soil. When adequate moisture and temperatures are maintained, oxidation of elemental S will be accomplished within one or two growing seasons. Sulfuric acid is NOT recommended due to the immediate dangerous reaction that occurs and creation of excessive salts that can lead to poor plant growth. Some growers apply sulfuric acid to lower soil pH, but it is expensive and only effective in the upper inch or so of the soil. Other liquid forms of acid are also not recommended because of the increase in soil salinity after the reaction has taken place.
Saline and Sodic Soils
Soils that are both saline and sodic are the hardest to manage. By definition, these soils have an EC above 4 mmhos/cm and an ESP greater than 15%. These soils must first be managed as sodic soils. Application of calcium to disperse the sodium followed by leaching to remove salts is critical. High EC water is beneficial to sodic soils for leaching purposes but is not beneficial for crop production. The soil must be irrigated after the application of calcium to remove the sodium that has been displaced by the calcium. Water is applied to leach the sodium to below the root zone of the plant, and water must therefore be free to drain or leach below the root zone.
Saline and sodic soils are often caused by natural factors. No amount of reclamation will make these soils satisfactory for crop production. The best that can occur is to use tolerant vegetation and maintain soil cover to keep the soil from eroding by wind or water. Often, plants that are native to alkali or salty areas are best suited for these saline-sodic soils. Consider using halophytes or alkali grasses and shrubs to get ground cover established.
Sources of Salinity and Sodium
The earth is composed of minerals of all kinds. Due to the effects of water and other weathering factors, minerals are dissolved and relocated somewhere else. If the water table is shallow, salts rise to the surface with water, and as the water evaporates the salts are left behind. Over geologic time, a lot of minerals have been dissolved in our soils and transported out to sea; this is why our oceans are salty. However, not all areas receive enough rain to wash the salts away or leach them out of the soil. These are natural occurrences, but they can be exacerbated by human activity and may create problems for farming or gardening.
Fertilizers are minerals that have value to plants as nutrients. These minerals usually have a high degree of solubility and can contribute significantly to the salinity of the soil if applied at heavy rates. Routine fertilization that takes into account soil levels of nutrients usually prevents the gross over-application of mineral fertilizers. Organic amendments, however, are more easily over-applied because many people are after the benefits of the carbon contained in the amendment. Manures and composted manures from livestock can have significant fertilizer value for plants due to the fact that supplemental minerals are often included in the animal's diet; however, these amendments are often very salty and should be managed carefully. Failure to account for these minerals in organic amendments from livestock sources often leads to excess salinity in the soil.
Adding irrigation water from surface or groundwater supplies leads to an increase in soil salinity. Ground and surface water are in direct contact with minerals, and there is always some degree of dissolution of those minerals into the water. The saltiness of any given water used for irrigation depends on the geology of the aquifer and the land through which it passes. It is always a good idea to have irrigation water evaluated for salinity and sodium levels.
Soil Salinity and Sodium Concerns Across New Mexico
Approximately 19% of the soil samples received by NMSU's SWAT lab from 2000 to 2008 had an EC greater than 4 mmhos/cm (Figure 4) and are considered saline. All areas of the state are subject to sources of salts as described in the previous section. Approximately 18% of the samples submitted from 2000 to 2008 had ESP levels above 6% (Figure 5). However, approximately 53% of the samples classified as saline had an ESP greater than 6% where a calcium amendment would be suggested.
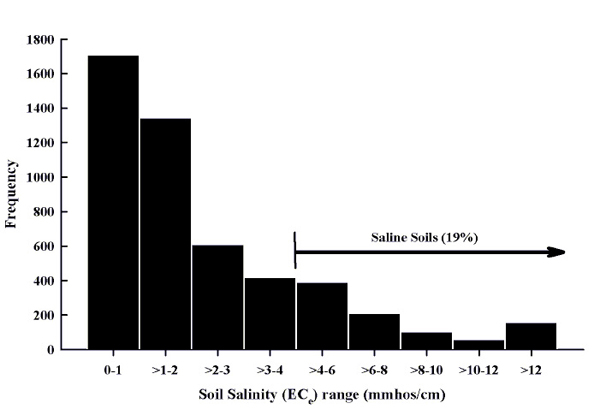
Figure 4. Soil salinity histogram for soils submitted to the NMSU Soil, Water, and Agricultural Testing Lab, 2000 to 2008.
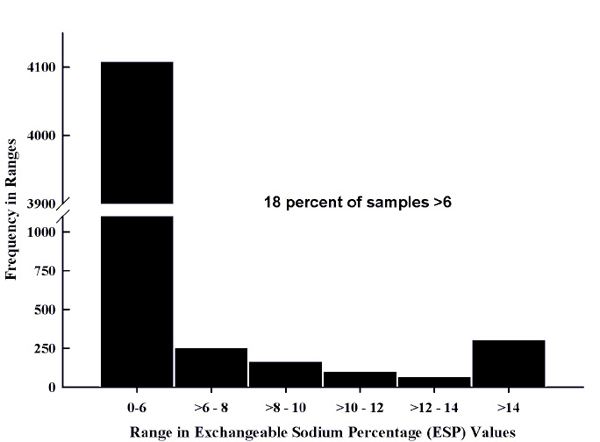
Figure 5. Range in exchangeable sodium percentage (ESP) values for samples submitted between 2000 and 2008.
It is always better to have your soil tested than to assume that you do, or don't, have a salinity or sodium problem. NMSU's SWAT lab uses the standard procedures explained in this publication as part of their routine analysis of soils. You are encouraged to submit soils to a laboratory of your choice, but always request a salinity assessment that follows standard procedures for assessing saline and sodic soils.
References
Kamphorst, A., and G.H. Bolt. 1978. Saline and sodic soils. In G.H. Bolt and M.G.M. Bruggewert (Eds.), Soil chemistry. A. Basic elements, 2nd ed (pp. 161-166). Amsterdam: Elsevier Scientific.
Maas, E.V. 1986. Salt tolerance of plants. Applied Agricultural Research, 1, 12-26.
Rhoades, J.D., A. Kandiah, and A.M. Mashali. 1992. The use of saline waters for crop production [FAO Irrigation and Drainage Paper 48]. Rome: Food and Agriculture Organization of the United Nations.
Glossary
Acre-inch: Usually used when referring to irrigation water quantities delivered to a field. One acre-inch is the depth of water needed to cover 1 acre, and is equivalent to 27,154 gallons of water.
Anion: A negatively charged ion such as chloride (Cl-), sulfate (SO42-), carbonate (CO32-), or bicarbonate (HCO3-).
Cation: A positively charged ion such as calcium (Ca2+), magnesium (Mg2+), sodium (Na+), potassium (K+), ammonium (NH4+), and others.
Cation Exchange Capacity (CEC): The degree to which soil can absorb and exchange cations.
Electrical Conductivity (EC): The ease with which electrical current passes through water. EC is proportional to the salt concentration in the water. Consequently, total salt concentration in a soil or irrigation water can be estimated by measuring EC. The higher the EC, the greater the salt concentration. ECe refers to the soil EC determined from a saturated paste extract. ECw refers to the EC of an irrigation water sample.
Elemental sulfur (S): A yellow, inert crystalline mineral that is finely ground. In soil, elemental S is oxidized to sulfate via microbial activity. The rate of elemental S oxidation in soil is most rapid in warm, moist soils. Complete oxidation of elemental S to sulfate often takes one to several years.
Exchangeable sodium percentage (ESP): Percentage of the cation exchange capacity that is filled by sodium. It is calculated as:
ESP = ((Na+, meq/100 g soil) / (CEC, meq/100 g soil)) × 100
To convert SAR to ESP, use the following equation:
ESP = (1.475 × SAR) / (1 + (0.0147 × SAR))
Gypsum: CaSO4•2H2O, the common name for calcium sulfate. It is applied as a source of calcium to reclaim sodic and saline-sodic soils.
Gypsum requirement (GR): The approximate amount of gypsum needed per acre to lower the ESP of the soil to a desired level. It is calculated as:
GR (ton/ac) = (present ESP – desired ESP) × CEC × bulk density × depth × 3.84
The factor of 3.84 assumes CEC is in meq/100 g or cmol(+ charge)/kg soil units. If CEC is in mmol(+ charge)/kg soil, the factor is 0.384. These factors assume 100% reclamation efficiency and a desirable SAR (<6) in the irrigation water. Always divide the calculated ton/ac by the percentage gypsum in the amendment to arrive at the correct amount to add.Leaching requirement: The leaching fraction necessary to keep soil salinity, chloride, or sodium (whichever is the most limiting factor) from exceeding a tolerance level for the crop rotation. Leaching requirement refers to long-term average conditions.
meq/L: Milliequivalents per liter. An equivalent is a term used in chemistry where the molecular weight of an ion is divided by its charge. The molecular weight of sodium is approximately 23 and the charge on a sodium ion is +1. Dividing the molecular weight by its charge gives an equivalent weight of 23. Calcium has a molecular weight of 40 and has a +2 charge, so the equivalent weight is 20.
pH: A measure of the acidity or basicity of a material or solution. Below 7 is acidic, above 7 is basic, and 7 is neutral. The pH is measured with a pH electrode and meter or dyes.
ppm: Parts per million. Also expressed as mg/kg in a solid matrix or mg/L in solution.
Relative yield: Actual crop yield divided by potential crop yield. Can be reported as a fraction or multiplied by 100 and reported as percent. Yield can refer to plant dry matter or harvestable economic material.
Saturated soil paste: A reference-state mixture of soil and water used for measuring EC, SAR, and pH. At saturation, the soil glistens slightly as it reflects light, flows slowly when the container is tipped, and slides freely and cleanly from a spatula.
Sodium adsorption ratio (SAR): The SAR of a saturated paste extract or irrigation water is a relationship between the concentrations of sodium (Na+) and calcium plus magnesium (Ca2+ + Mg2+). SAR reflects the Na+ status of the soil cation exchange capacity. It is calculated as:
SAR = ([Na+] / (0.5 ([Ca2+] + [Mg2+]))1/2 )
where calcium, magnesium, and sodium concentrations are expressed in units of milliequivalents per liter (meq/L).
Robert Flynn is an Associate Professor of Agronomy and Soils and an Extension Agronomist at New Mexico State University. He earned his Ph.D. at Auburn University. His research and Extension efforts aim to improve grower options that lead to sustainable production through improved soil quality, water use efficiency, and crop performance.
To find more resources for your business, home, or family, visit the College of Agricultural, Consumer and Environmental Sciences on the World Wide Web at pubs.nmsu.edu
Contents of publications may be freely reproduced for educational purposes. All other rights reserved. For permission to use publications for other purposes, contact pubs@nmsu.edu or the authors listed on the publication.
New Mexico State University is an equal opportunity/affirmative action employer and educator. NMSU and the U.S. Department of Agriculture cooperating.
Printed and electronically distributed August 2011, Las Cruces, NM
