Circular 607
Joe Ellington, Professor, Department of Entomology, Plant Pathology, and Weed Science (EPPWS)
Tracey Carrillo, Senior research specialist, EPPWS
John White, Doña Ana County Extension Agent-Agronomy and Horticulture/4-H
Carol Sutherland, Extension Entomologist/State Entomologist, Department of Extension Plant Sciences
David Richman, Science Specialist, EPPWS
Jeff Drake, Research Scientist, USDA/APHIS
Scott Bundy, Assistant Professor, EPPWS
College of Agriculture, Consumer and Environmental Sciences New Mexico State University (Print Friendly PDF)
Introduction
Sometimes insects, mites or their relatives limit enjoyment of our landscape and garden plants. We are familiar with pests that infest fruit, vegetables, flowers and foliage or make plants wither, change colors, develop spots or streaks or even die. But just as pests damage some plants, those same pests probably have one or more natural enemies that limit their numbers and their damage.
“Biological control” of pests involves either natural or human-assisted control of certain pest species by predators, parasites (parasitoids, as defined below) or pathogens. This circular outlines important definitions, concepts and examples of biological control of some common yard and garden pests in New Mexico. Home gardeners should note that biological control is complex, and they should assess the pros and cons of adopting these techniques as part of an overall Integrated Pest Management (IPM) strategy for their properties.
Not all insects and their relatives are harmful to landscapes. Relatively few species cause most of the damage to plants. The diversity of plants in most home landscapes tends to limit the damage done by any particular pest. Through observation, reading and short courses on the subject, gardeners can learn to recognize those species that are landscape pests and, perhaps, even some species that are considered beneficial. Some of the less common and smaller pest species and their natural enemies may offer special challenges for identification and management. But further study, experience and observation can help in manipulating them.
In IPM, a variety of pest control measures are used to reduce pest populations below damaging levels. Many tools are available, but some will be more valuable, manageable, available or affordable than others. Some tools for home gardeners include:
- Sanitation-cleaning: Remove or destroy debris if it is infested, or harbors pests. Sometimes leaving infested debris is an option, if it harbors beneficial insects.
- Physical-mechanical control: Hot or cold treatments, crushing, burying, screening or covering affected plants to prevent infestation.
- Cultural control: Rotate crops, till soil, try irrigation strategies, trap crops, or adjust planting and harvesting dates.
- Genetic control: Use plant varieties bred for resistance to certain pests.
- Chemical control: Use noninvasive inorganic or organic chemicals to kill or manage pests.
- Biological control: Conserve natural enemies already present in the area, introduce new species or augment existing populations of natural enemies (parasitoids, predators or pathogens) to decrease populations of particular pests below damaging levels.
Management Practices that Enhance Biological Control
Biological control is a broad concept that can be used to manage pests on many kinds of plants, including annual bedding plants, vegetables, and perennial flowers, shrubs, vines and trees. In practice, biological control of particular pests may be implemented by conserving the various species of natural enemies already present in the area. Where no effective natural enemy is present, entomologists or various government agencies may introduce one or more new species to the area. Candidates for release are screened carefully before permits are issued for release to ensure that the introduced species don’t become pests themselves. These introduced species may be released into infested areas immediately after screening, or they may be lab reared through several generations to increase their numbers before release. In some cases, entomologists may recommend augmentation as another means of enhancing biological control of a pest. In these situations, the natural enemy species is already present, but its numbers are too low to reduce a pest population below damaging levels. In response, entomologists may rear that natural enemy species through several generations so that, after release, its numbers are higher and the species has a better chance of controlling the pest. Here are additional ideas that may enhance biological control:
-
- Reduce or eliminate insecticides, especially those that are nonselective or have long residuals.
- If insecticides are used, apply them in noninvasive ways.
– Apply when beneficial insects are not present.
– Apply reduced rates and only when warranted (not calendar sprays or “preventative treatments”).
– Apply to alternate rows or alternate plants or only to affected plants.
– Apply systemic or host-specific insecticides.
– Apply to specific restricted areas of plants, such as trunks or roots.
- Provide sources of nectar, pollen and alternate hosts for beneficial insects during the growing season.
- Provide overwintering sites for beneficial insects.
- In general, don’t kill all of the harmful insects in an ecosystem. Reducing the density of harmful species, in host-specific ways, often is acceptable. This practice allows the beneficial complex to continue building for future control. The longer you can refrain from applying an insecticide, the more likely biological control will work.
Selected chemicals have a place in certain yard and garden IPM programs, because they are considered less toxic to beneficial insects and mites due to the ways they are applied or their modes of action. Broadly labeled insecticides, which are persistent and generally toxic to a wide variety of insects, can be harsh to garden ecosystems, resulting in environmental contamination, possible increased insecticide resistance in target pests, decreased beneficial insects, a resurgence of target pests, increased prominence of secondary pests and their damage, and potential damage (phytotoxicity) to plants that required protection in the first place.
Some products, such as pheromones, are host specific, impacting just one or a few species. Many pheromones are sex attractants, analogous to a perfume that aids one sex in locating the other as well as releasing additional courtship behavior. In commercial catalogs, pheromone products are named according to the species they attract. These blends of organic chemicals usually attract only one sex. For most moths, males are attracted to chemical baits that resemble pheromones produced and released naturally by females of the species when they are ready to mate. Because some similar insect species may be attracted to sticky traps because of their color, placement or pheromone mixture, make sure the insects on the traps are the target pests, rather than harmless look-alikes. While trapping might seem ideal for eliminating insecticides, it is more the exception than the rule. However, it indicates how detailed studies of pest biology and behavior can be useful in developing pest control protocols. Synthetic sex pheromones for adult moth attraction can be used in sticky traps to monitor flights and predict egg laying or hatching times. If a pesticide must be used to control certain pests, plotting the pest captures in pheromone traps can be helpful to properly target treatments. More recently, entomologists have found that “over dosing” some commercial crops with certain pheromones confuses at least a few pest species to the point that males can not find females for mating. This strategy continues to be studied as a tool for commercial agriculture. For the homeowner, currently registered pheromone products are labeled for pest detection and tracking rather than control. Always read and follow label directions on these products.
Bacillus thuringiensis (Bt) toxins can be specific to particular insect groups. This naturally occurring bacterium has been long known to kill certain kinds of insects. The bacterial infection of certain caterpillars, for example, turns lethal when the bacteria release toxins into the insect gut, paralyzing and killing it. Bt can be grown in cultures and commercially produced for homeowner use in specific applications. There are several subspecies of this bacterium, each affecting different kinds of insects. Subspecies kurstaki, the longest recognized and commercially produced Bt product, affects caterpillars or “worms” of certain butterfly and moth species. Subspecies israelensis affects certain aquatic fly larvae, especially mosquitoes and black flies. A third subspecies, sandiego or tenebrionis, kills certain beetle larvae or grubs, such as those of elm leaf beetles. For such products, homeowners need to read the labels carefully not only for application methods but also for limitations. Properly identifying pests susceptible to these treatments is a must.
Elemental sulfur can be used to control spider mites in some situations. Sulfur also can cause phytotoxicity in cantaloupes and cucumbers. However, sulfur does not seem to have direct negative effects on nontarget organisms.
Insect growth regulators (IGRs) are compounds that alter insect growth patterns in various ways that result in metabolic errors and asynchronous development. They are species- or group-specific and have a low toxicity to other life forms. They are recommended for garden and home use when possible.
Other products can be effective against a wide variety of insects. Examples include azadirachtin (neem), malathion, and carbaryl (Sevin®). These products vary considerably in toxicity to nontarget organisms depending on dose, application mode and amount of area treated. Some newer systemic insecticides (for example, Imidacloprid) are effective against insects but not mammals when used as directed on the label. Insects not feeding on or in treated tissues will have little chance of contacting the toxin.
Insecticides formulated as granules typically are applied to the soil, specifically, in the plants’ root zones where they have little contact with nontarget organisms resting on other parts of treated plants. Granular materials often have additional application requirements, such as watering or incorporating into the soil.
Using pesticides may be warranted in cases where swift, relatively inexpensive, reliable and highly effective control is required to protect a crop or special planting from being damaged or destroyed. Home gardeners may have various reasons for not relying solely on pesticides including expense, hassle of application, cleanup, disposal and any potentially negative effects on the health of people, pets, livestock or plants. Also, broad spectrum insecticides might reduce beneficial insect populations. Thus, in an IPM framework, home gardeners may be very interested in learning more about biological control and manipulating beneficial organisms in sustainable ways to help decrease pest problems on their properties. Insecticides and miticides that are relatively nontoxic to beneficial insects are listed in Table 1.
Table 1. Insecticides and miticides that are relatively nontoxic to beneficial insectsb
| Pesticide Chemical Name, Examples | Typea | Target Pests |
| Azadirachtin, neem products (Azatin®) | B | Insects and mites |
| Bacillus thuringiensis spp., including three subspecies, as follows: kurstaki (Dipel®, Thuricide®) israelensis (Bactimos®, Gnatrol®) sandiego or tenebrionis (M-Trak®, Novodor®) |
M M M |
Caterpillars Mosquitoes, some flies Leaf beetles (e.g. elm leaf beetle) |
| Tebufenozide (Confirm®) | IGR | Caterpillars (e.g. pecan nut casebearer) |
| Copper Bands | CON | Snails, slugs |
| Enstar II® | IGR | Whiteflies, aphids, scales |
| Esfenvalerate (Ortho Bug-B-Gone®) | CON and S | Insects |
| Horticultural oil (both “summer” and “winter”) | CON | Insects and mites |
| Imidacloprid (Admire®, Marathon®) | S | Whiteflies, aphids |
| Insecticidal soap (M-Pede®, Ringers®, Safers®) | CON | Aphids, mites |
| Lime sulfur | CON | Mites |
| Malathion | CON | Insects and mites |
| Metaldehyde (Deadline®) | CON | Slugs and snails |
| Permethrin | CON | Insects (and others) |
| Pheromones (numerous commercially available products) | P | Many species of moths (e.g.codling moth, pine-tip moths) |
| Carbaryl (Sevin®) | CON | Insects |
| Spinosad (Conserve®, Success®) | M | Caterpillars |
| Sticky materials (Tanglefoot®, Stickem®) | CON | Trunk climbing insects |
| Sulfur | IC | Mites |
| aKey: B, botanical; M, microbial; CON, contact; S, systemic; P, pheromone; IC, inorganic contact; IGR, insect growth regulator. bSome of the listed labeled controls have not been studied thoroughly in New Mexico. New Mexico State University is not responsible for use or misuse of these products. This list is subject to change. Consult your County Agent to determine appropriately labeled and currently registered pesticides. |
||
Some pesticides are safe, when applied as per label directions, to warm-blooded animals but toxic when applied topically to beneficial insects. Esfenvalerate, malathion, carbaryl and metaldehyde fall into this category. Metaldehyde is the active ingredient in snail and slug bait. The other three pesticides are organic contact materials used against various insects and some mites.
Chemicals can have a place in managing yard and garden pests, if they are used judiciously and appropriately. Always read the complete label for any of these products and follow instructions completely.
Types of Natural Enemies, Common Yard and Garden Pests and their Taxonomy
All insects are not damaging to plants, and not all damaging insects affect all plants. While some pests might affect only one plant, others can damage several to many kinds of plants. Pecan nut casebearer, a pecan pest, is an example of the former, while green peach aphid, a pest of many species of ornamental, vegetable and stone fruit tree species, is a good example of the latter. Further, some pests can have several generations each growing season. For some hosts, pest damage can be cumulative, taking place over one or more growing seasons.
The tables that follow list some specific insect pests of garden and landscape plants, along with certain natural enemies that attack them. Natural enemies may be predators, parasitoids or pathogens.
- Predators include insects, spiders or mites that kill more than one prey item during their life cycles. Many predatory species have unique hunting behaviors and habitat preferences that help them find and kill their prey. Some species are predators in one life stage but not another, while others are predatory in all but their egg stage. Many species of predators are “generalists”, feeding on a variety of pest species. Some predators, of great significance to biological control, kill hundreds or thousands of prey in their lifetimes and are prolific breeders.
- Parasitoids are insects that kill one host as a consequence of their development. Adult female parasitoids have unique habitat and hunting behaviors that permit them to find specific host species or a group of host species that they use as a place to deposit eggs. Depending upon species, female parasitoids lay eggs either on or inside the host; the developing larva gets all of its food from that host individual (either inside the host as an endoparasite, or outside the host as an ectoparasite). The emergence of the mature larva or the adult for some species, kills the host. The adult parasitoid may feed on plant nectar, honeydew, host insect blood or nothing at all, subsisting on stored fat. In contrast, fleas and lice are good examples of true parasites. Usually, these pests feed sporadically on their hosts, but do not kill them as a result of their feeding or reproduction.
- Pathogens include various disease-causing microorganisms, especially fungi, bacteria, viruses and similar microscopic life forms. Some fungi may affect numerous species of hosts, while some bacteria produce toxins affecting only certain groups of hosts. For example, one strain of Bacillus thuringiensis bacteria produces one toxin that affects only certain caterpillars, while another strain’s toxin affects only certain beetles or aquatic fly larvae.
Before proceeding, a longer explanation of taxonomy is in order, especially for gardeners who use insect identification handbooks or intend to order or understand many species of biological control agents.
In the hierarchy of taxonomy, the Phylum Arthropoda is the most inclusive category while the species is the narrowest, most exclusive.
Phylum Arthropoda—All organisms that lack backbones, have external skeletons and jointed legs (the literal translation of “arthro-pod”) belong to this very large and diverse group. Arthropod can be a useful term to include insects and any appropriate group of their close relatives. In this publication, mites and spiders are two such groups. Mites contain both plant pest and predatory species in addition to many thousands of other species with diverse habits. Mites are wingless, eight-legged, very small to microscopic organisms with usually oval bodies, no antennae and paired mouthparts. Spiders belong to another group of arthropods discussed below as predators. These also are wingless, eight-legged creatures that range in size from barely visible with the naked eye to the size of a tarantula. Most of the common species in our area are smaller than a quarter in diameter, only a few have the physical ability to bite man, and of those, only two are considered medically significant (black widow and violin spiders, including the brown recluse).
Class Insecta—Adult insects generally have a pair of antennae, three pairs of legs, three body regions and two pairs of wings (although there are numerous exceptions to the latter).
Order—Examples of eight insect orders are discussed in this publication. Many order names have the suffix ‘-ptera,’ meaning wing. These include:
Neuroptera (nerve-winged insects, the lacewings)
Diptera (two-winged insects, the “true” flies)
Hymenoptera (membrane-winged insects, including the small to minute parasitic wasps and numerous other species)
Coleoptera (sheath-winged insects, the beetles)
Hemiptera (half-winged insects, the “true” bugs) and (alike- or uniform-winged insects, including aphids, scales, whiteflies, leafhoppers and numerous other species). Note: In recent books, Homoptera are combined under Hemiptera.
Lepidoptera (scaly-winged insects, moths and butterflies)
Thysanoptera (fringe-winged insects, the thrips)
Family—These capitalized names end in “-idae;” when used descriptively, the family name will be in lower case letters, ending in “id”. Example: Coccinellidae is the full family name for the lady beetles. To use the family name descriptively, refer to them as coccinellids or coccinellid beetles.
Genus—The first part of an organism’s scientific name capitalized and either underlined or in italics (see below).
Species—The second part of an organism’s scientific name is in lower case letters and is either underlined or in italics (see below).
Author—the third part of a scientific name may not always be shown. This is the last name of the person who originally described and published the species, placing it in a particular genus. If an author’s name is in parentheses, that person originally described the species in a different genus than where it is presently placed. Sometimes, two (rarely more) authors are responsible for describing and publishing a species name. Author names have been omitted in this publication. Frequently, they also are omitted from the biological control catalogs.
An example of a scientific name is Tetranychus urticae Koch. The common name for this organism is two-spotted spider mite.
At times, scientists may refer to more than one species in a genus by using the abbreviation ‘spp’. In other cases where the species is either not identified or not described, the abbreviation ‘sp.’ may be used. As examples, consider one member of a beneficial wasp genus as Encarsia sp., while Olla spp. would refer to two or more species of this ash-gray lady beetle.
Many insects and mites in tables 2-18 are listed by their “common names.” Insects or mites may have none, one or more than one common name which may vary from one part of the country to another. Common names may not be precise in describing a specific organism, its hosts or roles in the environment. Entomologists and other scientists often use scientific names to be more precise. Rooted in Greek or Latin, scientific names consist of a genus, species and author names. While all described species of insects and mites have scientific names, relatively few have common names. Therefore, in various literature and biological control catalogs, interested gardeners will have to consider scientific names carefully.
Some Specific Arthropod Pests of Garden and Landscape Plants with Biological Control Alternatives
Mites
Most pest mite species are nearly microscopic (Table 2). They are wingless relatives of insects and have only two body regions that are more or less combined into one, no antennae, and four pairs of legs as nymphs and adults. Spider mites are notorious pests of roses, many vegetable and garden annual species and occasionally trees. Their populations typically soar during hot, dry weather and are extremely difficult to manage under outbreak conditions. Mites that cause certain plant galls and blisters are microscopic and worm-shaped. Many species are host specific and cause characteristic plant growth abnormalities, particularly galls. Several species of predatory mites (Table 10), thrips and some true bugs (eg. big-eyed bugs and minute pirate bugs) feed on plant eating bugs.
Table 2. Mites that are potential plant pests and some of their natural enemies.a
| Specific Pest | Arthropod Predators Active on a Variety of these and other Mites |
| Tetranychus spp. (Figure 1) including web spinning spider mites, European red mites | Various predatory mite species, Table 10big-eyed bugs* (predators), Figures 42, 43 lacewings* (predators), Figures 50, 51, 52 midge larvae (predators) six spotted thrips* (predator) spider mite destroyer lady beetle (predator) western flower thrips (predator) damsel bugs (predator), Figures 44, 45 |
| *An asterisk after the genus means it is available commercially. aListed beneficials are indigenous, introduced into New Mexico or available from commercial sources. |
|
Thrips
A variety of predatory mites (Table 10), thrips and true bugs i.e. big-eyed bugs and minute pirate bugs feed on phytophagous mites. These agile, minute insects may be beneficial, predators or they may slash tender cells of their host plants, sucking up the fluid contents (Table 3). As predators, they may feed on insect or mite eggs or very young immature stages, even of their own species. As plant pests, their feeding damage often appears as “tiny cat scratches” on foliage, yellowing or browning areas or brown-edged, tattered flower petals on plants such as roses. Several species of thrips are capable of transmitting tomato spotted wilt virus.
Table 3. Thrips that are potential plant pests and some of their natural enemies. Listed beneficials are indigenous, introduced into New Mexico or available from commercial sources.
| Specific Pest | Relatively Host-specific Natural Enemies |
| Greenhouse thrips | Scolothrips sexmaculatus, six-spotted thrips and Leptothrips mali, black hunter thrips. Both are entirely predaceous species. |
| Western flower thrips, Figure 2 | Thripobius semiluteus (nymphal endoparasitic wasp) |
A variety of predatory natural enemies are active on different species of thrips including minute pirate bugs (Figures 40, 41) and certain mites. Western flower thrips are usually pests, but they also can be opportunistic predators and even cannibalistic. Some of the predatory mites and certain species of minute pirate bugs are available commercially.
Aphids
Aphids are tiny, globose, soft-bodied insects with piercing-sucking mouthparts that suck sap from their host plants (Table 4). They can cause host plants to wilt, resulting in foliage loss and premature color change. Some species also are carriers of certain plant viruses. Their dense colonies can be unsightly and their waste products, droplets of honeydew, can make affected foliage and surroundings sticky and shiny. Honeydew coated foliage may be visited by flies, bees, wasps, ants, and even small mammals. The affected host plant also could be subject to growth of damaging molds.
Table 4. List of common aphids and some of their natural enemies. Listed beneficials are indigenous, introduced into New Mexico or available from commercial sources.
| Specific Pest | Relatively Host Specific Natural Enemies** |
Natural Enemies that Attack these and a Variety of other Aphids and other Insect Prey |
| Asparagus aphid | Trioxys brevicornis (endoparasitic wasp) |
true bugs* (predators), Figures 40-49. earwigs (predators) lacewings (predators), Figures 50-52. lady beetles* (predators), Figures 55-61. syrphid flies (predators), Figures 53-54. spiders (predators), Figures 32-39. Entomophthora aphides (pathogenic fungus) Aphid mummy resulting from parasitization, Figure 64. |
| Bean aphid | Diaeretiella* spp (endoparasitic wasp) |
|
| Black bean aphid |
Lysiphlebus spp.* |
|
| Cabbage aphid | Diaeretiella rapae* (endoparasitic wasp) |
|
| Cotton or melon aphid | Lysiphlebus testaceipes* (endoparasitic wasp) |
|
| Corn leaf aphid | Lysiphlebus testaceipes* (endoparasitic wasp) |
|
| Green peach aphid, Figure 3 | Aphelinus* semiflavus (endoparasitic wasp) Aphidius matricariae* (endoparasitic wasp) Diaeretiella rapae* (endoparasitic wasp) Lysiphlebus testaceipes* (endoparasitic wasp) |
|
| Oleander aphid | (none known) | |
| Pea aphid | Aphidius* ervi (endoparasitic wasp) Aphidius smithi (endoparasitic wasp) |
|
| Rosy apple aphid | Lysiphlebus testaceipes* (endoparasitic wasp) |
|
| Wooly apple aphid | Aphelinus mali (endoparasitic wasp) |
|
| Yellow pecan aphid, Figure 4 | Trioxys pallidus Aphelinus* spp. |
|
| Black margined pecan aphid, Figures 5, 6 | Harmonia axyridis (predator) Trombidiidae (predaceous mite) Deraeocoris nebulosus |
|
| Black pecan aphid, Figures 7, 8 | (none known) | |
| *An asterisk means the genus or species is available commercially. **The relationship of the natural enemies to the specific pest is described within the parenthesis. An endoparasitic wasp inserts one or more eggs into its host. The parasitoids develop within their hosts, feeding on host tissues, eventually killing them. The immature wasp generally pupates inside its host, cutting a round hole through the host integument. The “dry shell” of the dead aphid is called a “mummy.” |
||
A variety of natural enemies attack many species of aphids but also take other insect prey. These include certain predatory true bugs (Figures 40-49), earwigs, lacewings (Figures 50-52), lady beetles (Figures 55-61), syrphid flies (Figures 53-54), and spiders (Figures 32-39). At least some species of predatory true bugs, lacewings and lady beetles are available from commercial sources; remember that some beneficial species are better suited to particular habitats and host species than others. Other beneficial species are indigenous or established in New Mexico after having been introduced here.
Whiteflies
Adult whiteflies (Table 5) are minute, white, moth-like insects with piercing-sucking mouthparts that billow from infested foliage like dandruff. The immature stages also have piercing-sucking mouthparts but are flattened and immobile like miniature scale insects. Some species, such as silverleaf whitefly, can transmit various plant pathogens while feeding. Large populations of these pests can develop rapidly on aging plants, hastening defoliation and plant death. Much of the damage done by these pests is similar to that of aphids, which are near relatives.
Table 5. Whiteflies that are potential plant pests and some of their natural enemies. Listed beneficials are indigenous, introduced into New Mexico or available from commercial sources.
| Specific Pest | Relatively Host Specific Natural Enemies |
| Ash whitefly | Clitostethus arcuatus (predatory lady beetle) Encarsia inaron (nymphal-pupal endoparasitic wasp) |
| Greenhouse whitefly, Figure 9 | Encarsia formosa* (nymphal-pupal endoparasitic wasp) |
| Silverleaf whitefly | Encarsia spp.* (endo- and ectoparasitic wasps) Eretmocerus spp.* (endo- and ectoparasitic wasps) Delphastus pusillus (predatory lady beetle) |
| *An asterisk means the genus or species is available commercially. | |
Natural enemies active on a variety of whitefly species include the following predators: big-eyed bugs (Figures 42-43), lacewings (Figures 50-52) and lady beetles (Figures 55-61). At least some species of each of these groups are available commercially.
Leafhoppers
Leafhoppers are small, wedged-shaped insects that suck juices from plants. Some transmit plant diseases, while others interfere with the plant’s normal physiology. Adults often run sideways and have one or more rows of spines on the hind tibia (second major segment of the back leg). Leafhoppers feed on a wide variety of plants, including trees, shrubs, and field and garden plants. One example is the grape leafhopper, a pest of grape foliage. It does have an effective egg and larval parasitoid in the tiny, endoparasitic wasp, Anagrus epos. This wasp is not available commercially, but does occur at least in some parts of New Mexico.
Scale Insects and their Relatives
Hard scales, soft scales and mealybugs belong to the insect order Homoptera (whole wing insects). Hard scale females are wingless, legless, eyeless degenerate insects covered by a hard round, oval or elongated shell. Soft scale females are covered with a thin shell, while mealybugs are dusted all over with a white, waxy powder. They often have short radiating threads around the margins of their bodies and occasionally two longer “tails”. Female hard and soft scales are motile during the first stage of development after hatching from eggs or being born alive. After the first molt, they lose their legs and become sessile, sucking sap from various parts of host plants. Mealybug females remain mobile their entire lives. The adult males of all species are very small gnat-like creatures that are sometimes wingless or are provided with a single pair of wings. These males often lack mouthparts and live only long enough to disperse and mate.
Several species of lady beetles are important predators of a variety of scale and mealybug species. Certain lady beetle species are available commercially.
Table 6. Scales and mealybugs that are potential plant pests and some of their natural enemies.*
| Specific Pest | Relatively Host Specific Natural Enemies |
| Cottony cushion scale, Figure 10 | Cryptochaetum iceryae (wasp parasitoid) |
| Apricot scale, Figure 11 Euonymus scale, Figure 12 |
Encyrtus californicus (wasp parasitoid) Hemisarcoptes malus (mite predator) Chilocorus kuwanae (lady beetle predator) |
| San Jose scale, Figure 13 | Cybocephaltus californicus (sap beetle) Chilocorus orbus (lady beetle predator) Aphytis spp. (nymphal-adult parasitic wasp) Encarsia perniciosi (endoparasitic wasp) |
| Oleander scale, Figure 14 | Aphytis spp. |
| Mealybugs, Figure 15 | Allotropa spp. (wasp parasitoid) Pseudaphycus spp. (wasp parasitoid) Tetrachemoides spp. (wasp parasitoid) |
| * Listed beneficials are indigenous or introduced into New Mexico. | |
True Bugs
True bugs are members of the insect order Heteroptera (“different-winged” insects). The fore part of the wing is opaque and slightly leathery while the latter half is membranous. All true bugs have piercing-sucking mouthparts in both immature (nymph) and adult stages. All of our common species have simple metamorphosis, meaning the immature stages resemble miniature versions of the adult. Adults and nymphs have fairly large, multi-faceted compound eyes. Older nymphs of winged species have small, external wing buds. The life cycle proceeds from the egg stage, through a series of ever-larger nymphs and the adult. Only adults are capable of reproducing and, if winged, flight. Many species of true bugs defend themselves by releasing foul-smelling fluids from glands on their abdomens. Stink bugs are famous for smelling bad when roughly handled or crushed.
The true bugs listed in Table 7 are potential plant pests. Many other species exist in the home garden and landscape that do not affect plant health. Some bug species may be considered beneficial in that they prey on pest species of insects or weeds.
Table 7. True bugs that are potential plant pests and some of their natural enemies.a
| Specific Pest | Relatively Host Specific Natural Enemies |
| Bordered plant bug, squash bug | Trichopoda pennipes (adult endoparasitic fly) |
| Lygus bug, Figures 16, 17 | Anaphes iole (egg endoparasitic wasp) |
| Stink bugs, Figure 18 | Trissolcus basalis, T. euschisti (egg endoparasitic wasps) Gymnosoma fuliginosum (larval endoparasitic fly) Telenomus spp. (egg endoparasitic wasp) |
| aListed beneficials are indigenous, introduced into New Mexico or available from commercial sources. | |
Some natural enemies are active against a variety of true bugs. Several species of parasitic wasps may attack one or more life stages of true bugs. Predatory natural enemies include: assassin bugs (Figures 46, 47 and 48 big-eyed bugs (Figures 42-43), damsel bugs (Figures 44-45), predatory flies (Figures 53-54) lacewings (Figures 50-52), and spiders (Figures 32-39).
Beetles
Beetles listed in Table 8 are potential plant pests. The Colorado potato beetle and elm leaf beetle complete their life cycles above ground, primarily feeding on the foliage of their respective hosts. The masked chafer, May or June beetles, Japanese beetles and their relatives have winged adult stages that are active above ground in spring or summer. While most of these adult scarab beetles fly, mate and lay eggs at night, Japanese beetles and green June beetles do so during the day. Adult Japanese beetles skeletonize foliage and blossoms of numerous species of broadleaf plants. Green June beetles frequently attack ripening fruit. Other species of scarab beetles may not feed in the adult stage. The larval stages of all of these scarab beetles and other members of the scarab beetle family are commonly called white grubs. When numerous, these plump C-shaped, off-white grubs can cause serious damage to root zones of grasses and other shallow rooted plants.
Table 8. Beetles that are potential plant pests and some of their natural enemies.a
| Potential Pest | Relatively Host-specific Natural Enemies |
| Colorado potato beetle | B. thuringiensis ssp. tenebrionis* (pathogen) |
| Elm leaf beetle, Figure 19 | B. thuringiensis spp. tenebrionis* (pathogen) Oomyzus (Tetrastichus) gallerucae (egg, pupal endoparasitic wasp) |
| Masked chafer, May or June beetles, striped June beetle and other related species |
Heterorhabditis bacteriophora (entomopathogenic nematode) Steinernema glaseri (entomopathogenic nematode)-either of these predatory nematodes would be applied to the soil in areas infested with white grubs. The nematodes attack and kill the grubs in the soil. |
| *An asterisk means the genus or species is available commercially. aListed beneficials are indigenous, introduced into New Mexico or available from commercial sources. |
|
Many other species of beetles exist in the home garden and landscape that are beneficial or do not affect plant health. Various natural enemies are active on a variety of beetles including: various pathogens (certain bacteria, fungi and viruses), true bugs (predators, Figures 40-49), predatory or parasitic true flies (Figures 53-54), certain parasitic small wasps, and particular species of entomopathogenic nematodes. Certain of the bacteria, true bugs and entomopathogenic nematodes are available commercially.
Caterpillars
Caterpillars are the immature larval stages of moths and butterflies, sometimes called “worms” (Table 9). Most caterpillars have spherical or button-shaped heads that are easy to see and long, soft, cylindrical bodies. Most often, there are three pairs of short, jointed legs behind the head and several (up to five) pairs of fleshy legs on the abdomen. Caterpillars can be “bare”, fuzzy or spiny, depending upon age and species. Many species feed externally on plants, particularly on foliage, flowers, fruit or buds; some are leafminers, and still others bore into their plant hosts.
Table 9. Caterpillars that are potential plant pests and some of their natural enemies.a
| Specific Pest | Relatively Host Specific Natural Enemies |
Natural Enemies Active on these and a variety of other Caterpillars |
| Cabbage looper, Figure 20 | Copidosoma truncatellum (egg-larval endoparasitic wasp) Hyposoter exiguae (larval endoparasitic wasp) Microplitis brassicae (larval endoparasitic wasp) nuclear polyhedrosis virus* (pathogen) Trichogramma pretiosum* (egg endoparasitic wasp) Voria ruralis (larval endoparasitic fly) Chelonus insularis Cotesia glomerata |
Bacillus thuringiensis* (pathogen) big-eyed bugs* (predators), Figures 42, 43 birds (predators) damsel bugs (predators), Figures 44, 45 earwigs (predators) flies (predators), Figures 53, 54 ground beetles (predators), Figure 62 lacewings* (predators), Figures 50-52 minute pirate bugs* (predators), Figures 40, 41 nematodes* (entomopathogens) spiders (predators), Figures 38, 39 viruses* (pathogens) wasps (parasites) assassin bugs (predators), Figures 46-48 Deraeocoris nebulosus (predators) |
| Beet armyworm, Figure 21 | Euplectrus sp. | |
| Yellow-striped armyworm | Euplectrus sp. Hyposoter exiguae (larval endoparasitic wasp) Lespesia archippivora (larval-pupal endoparasitic fly) Chelonus texanus Campoletis sp. (larval endoparasitic wasp) Euplectrus comstockii Trichogramma spp.* (egg endoparasitic wasp) |
|
| Pecan nut casebearer, Figures 22, 23, 24, 25 |
Allothrombium sp. (predacious mite) Calliphialtes grapholithae Phaneratoma fasciata Lixophaga mediocris Macrocentrus instabilis Bassus acrobasidis (larval endoparasitic wasp) Brachymeria hammeri (larval endoparasitic wasp) Goniozus legneri * (larval exoparasitic wasp) Cremastus spp. (larval endoparasitic wasp) Clarkinella spp. |
|
| Sulfur butterfly, Figure 26 | Euplectrus sp. | |
| Codling moth, Figure 27 | Trichogramma spp.* (egg endoparasitic wasps) granulosis virus (pathogen) Mastrus ridibundus (larval-pupal endoparasitic wasp) |
|
|
Corn earworm, also known by |
Meteorus leviventris Meteorus autographae Rogas perplexus Bracon spp. Campoletis sp. Euplectrus sp. Chelonus texanus (= C. insularis) Chelonus insularis Hyposoter exiguae (larval endoparasitic wasp) Trichogramma spp.* (egg endoparasitic wasp) |
|
| Diamondback moth | Trichogramma spp.* (egg endoparasitic wasps) |
|
| Gypsy moth (not presently found in New Mexico) |
nuclear polyhedrosis virus* (pathogen) (wasps, including numerous parasitoid species) |
|
| Imported cabbageworm | Cotesia glomerata Trichogramma spp.* (egg endoparasitic wasps) |
|
| Nantucket pine tip moth | Trichogramma minutum* egg endoparasitic wasp Trichogramma platneri * (egg endoparasitic wasp) |
|
| *An asterisk means the genus or species is available commercially. | ||
As a group, caterpillars may be attacked by a wide variety of natural enemies including: Bacillus thuringiensis (pathogen), big-eyed bugs (predators, Figures 42-43), lacewings (predators, Figures 50-52), minute pirate bugs (predators, Figures 40-41), entomopathogenic nematodes, and certain viruses that affect only insects. Several species of each of these are available commercially. In addition, a variety of birds, damsel bugs (Figures 44, 45), earwigs, predatory true flies (Figures 53-54), ground beetles, spiders (Figures 32-39), assassin bugs (Figures 46-48) and the true bug Deraeocoris nebulosus (Figure 49) are known to prey on various species of caterpillars. Small parasitic wasps and parasitic flies also help suppress various caterpillar populations.
Predatory Arthropod Groups
Predatory Mites
Most predatory mites are pear-shaped, long legged, and shiny. Their mouthparts extend forward while the mouthparts of pest mites extend downward. Predatory mites can be very effective in controlling pest mites. Predaceous mites may feed on all stages of pest mites, scale crawlers, thrips and whiteflies. Outbreaks of pest mites often occur when broad-spectrum insecticides are used to kill other pests or when plants are water stressed.
Table 10. Examples of common predatory mites that are commercially available. Most species are for greenhouse use.*
| Mite Feeding Habits | Mite Species With These Habits |
| Feed on mites only | Phytoseiulus persimilis* Active at 70-80° F, some strains up to 100° F and 90% Relative Humidity (RH) Phytoseiulus longipes* Active at 40% RH and 70° F Feeds on mites almost exclusively |
| Prefer to feed on mites but also feed on pollen and other food |
Metaseiulus (=Galandromus) occidentalis (Western predatory mite) Active in hot climates and at least 50% RH Neoseiulus californicus* Active at 85–95° F. Needs RH of 65% |
| Generalized feeders on mites, insects, pollen, fungi and plant sap |
Amblyseius barkeri*, (= Neoseiulus) mites, feed on mites and thrips Euseius spp.*, shiny white mites, feed on small insects Hypoapsis* spp., reddish mites, feed on thrips and small insects |
| *An asterisk means the genus or species is available commercially. | |
Common Spider Families Found in Urban and Suburban Settings
Spiders are some of the best predators found in gardens and field ecosystems. They are relatives of insects that follow the same taxonomic hierarchy as described previously. They belong to the Class Arachnida and the Order Araneae.
Spiders have two separate body parts and eight legs unlike insects, which have three body parts and six legs. Spiders are classified as arachnids, along with mites. All spiders are predaceous and, as such, generally are beneficial, although some crab spiders may kill pollinators. There are some 3,500 spider species in the United States and some 500-1,000 species described in New Mexico. There are only two genera of spiders that are harmful to humans in the state, the widows and violin spiders, including relatives of the brown recluse spider. Although all spiders reduce insect populations, none are available commercially for release. All spiders are general feeders, but some are somewhat specific because of their habitat preferences. Because some wait for their prey in webs, they are more likely to catch adult flying insects than are ground-dwelling spiders. Alfalfa and other field crops are a good reservoir for many spider species and other beneficial insects. When alfalfa matures or is cut, beneficial insects and spiders disperse, looking for food and better habitat.
Guilds (groups of spiders that attack similar prey such as flying insects and caterpillars) are important in many agricultural systems. Table 11 lists guilds and examples found in urban and suburban landscapes and gardens.
Table 11. Some common spider guilds, examples of common species and notes on biology for selected spiders found in urban and suburban landscapes and gardens in New Mexico. These are indigenous or established species in New Mexico.
| Spider Family & Common Name | Examples of Common Species and Notes on Biology |
| Dysderidae (sowbug-eating spiders) | (specialists), also eat certain beetles, examples: Dysdera crocata, the European sowbug-eating spider, introduced from Europe. Fangs are elongated and used like ice tongs on large or hard-bodied prey. |
| Salticidae (jumping spiders, Figures 32, 33) and Oxyopidae (lynx spiders, Figure 34 ) | (active hunters), examples: Phidippus audax, Metaphidippus chera, Oxyopes salticus, Puecetia sp. |
| Lycosidae* (wolf spiders, Figure 35) | (runners and ambushers), examples: Pardosa sternalis, Hogna carolinensis |
| Tetragnathidae*, Araneidae or Uloboridae (orb-weavers, Figure 36) |
(trappers), examples: Tetragnatha laboriosa, Neoscona oaxacensis, Araneus illudatus, Uloborus sp. |
| Dictynidae, Lynyphiidae,* or Theridiidae (mostly space web weavers; theridiids also known as comb footed spiders, Figure 37) |
(trappers), examples: Dictyna personata, Latrodectus hesperus, Grammonota sp. NOTE: Latrodectus hesperus (western black widow, Figure 37) is venomous. |
| Miturgidae, Anyphaenidae or Gnaphosidae (sac and ground spiders, Figure 38) |
(runners), examples: Cheiracanthium inclusum, Hibana incursa, Herpyllus sp. |
| Thomisidae* and Philodromidae (crab spiders, Figure 39) |
(usually ambushers), examples: Misumenops sp., Misumenoides formosipes, Philodromus sp. |
| Mimetidae (pirate spiders) | (web invaders) obligate araneophages - (eat other spiders), example: Mimetus hesperus |
| * most abundant- individuals, making up over 90% of spiders found in Mesilla Valley alfalfa fields. | |
Common Terrestrial True Bug Families Known to Contain Predatory Species
There are approximately 37,000 species of true bugs known worldwide that feed on both plant and animal prey. The predatory true bugs listed here are a subset of the insect order Heteroptera as previously discussed. Predatory true bugs are characterized by having long, slender mouthparts that can pierce prey, inject digestive enzymes and suck up digested and liquified tissue. Having a simple metamorphosis, their life stages consist of eggs, a series of nymphs and adults. The nymphs look like miniature adults. Nymphs and adults have compound eyes, but nymphs lack wings and genitalia. The first pair of wings in adults is opaque, slightly thickened and stiff basally, while the distal or free end is thinner and usually membranous. A flattened triangular or shield-shaped structure lies between the bases of the front wings. True bugs (Table 12) may play an important part of nonhost-specific biological control in yard and garden settings in New Mexico. Some true bug species recently have become available from commercial insectaries.
Table 12. Common species of predatory true bugs with notes on their life cycles and habitats.a
| Family and Common Examples of Predatory Bugs | Notes on Life Cycles, Habitats |
| Anthocoridae, minute pirate bugs, Orius* spp., Figures 40, 41 | Orius feed on thrips, spider mites, aphids, lygus nymphs, insect eggs, newly hatched caterpillars and pollen. Adults are black with three triangular white markings. Eggs are inserted in plants along leaf veins. Nymphs are orange. |
| Geocoridae, big-eyed bugs, Geocoris* spp., Figures 42, 43 | Geocoris feed on insect eggs and small larvae; caterpillars, including tomato fruitworms; zhiteflies; mites, aphids; leafhoppers; and nymphs of true bugs. Eggs are laid on leaves, often in spider mite colonies. |
| Nabidae, damsel bugs, Nabis spp., Figures 44, 45 | Nabids feed on leafhoppers, aphids, true bugs and small caterpillars. Eggs are inserted in plant tissue. |
| Phymatidae, ambush bugs | Phymatids feed on bees, wasps, flies and other flying insects |
| Reduviidae, assassin bugs, Zelus and Sinea, Figures 46, 47, 48. | Reduviids attack nearly any insect prey they can handle. Eggs are laid in clusters on leaves. |
| Pentatomidae, stink bugs, Perillus, Podisus spp. | Most pentatomids are plant feeders, but some are important predators. The predators have spearlike mouthparts, which can point forward. They are oval or shield-shaped. |
| Miridae, predaceous plant bugs, Deraeocoris spp., Figure 49, Orthotylus sp. |
Plant bugs are among the most common insects. The majority are plant feeders, and some are destructive pests. Numerous species are predaceous. |
| *An asterisk means the genus or species is available commercially. aListed beneficials are indigenous, introduced into New Mexico or available from commercial sources. |
|
Lacewings
About 6,000 lacewing species are known worldwide. Except for a few brown species, adults are slender-bodied, green insects with large, green, prominently veined wings. Adults may be predaceous or pollen and nectar feeders. Larvae are spindle-shaped and covered with hairs and spines. Their bodies taper towards the head and tail. Larvae have sickle-shaped mouthparts, which are used to skewer and suck up their prey’s body contents. Most larvae are generalist predators, but some specialize by consuming small arthropods, especially aphids. Eggs are laid on slender hairs singularly or in groups. Pupae are housed in spherical, white, silken cocoons attached to tree trunks or other objects. Lacewing pupae may be heavily parasitized late in the season in New Mexico. Lacewings (Table 13) are available from commercial sources.
Table 13. Common species of lacewings with notes on their life cycles and habitats.a
| Lacewing Families and Examples | Notes on Lacewing Life Cycles and Habitats |
| 1. Chrysopidae - green lacewings | |
| Chrysoperla carnea*, Figures 50, 51, 52 Chrysoperla rufilabris* Chrysopa nigricornis |
Common in low-growing habitats. Adults feed on nectar and honeydew. More common in tree and shrub habitats. Adapted to more humid environments. Larvae are predaceous, but adults are not. Overwinter as adults. More aggressive as a predator. Common throughout the United States, prefers arboreal habitats. Adults are predaceous and require relatively high host density before laying eggs. Eggs are laid in groups. Overwinter in cocoons. |
| 2. Hemerobiidae - brown lacewings | |
| Hemerobius spp. (brown lacewings) | Eggs do not have stalks. Adults and larvae are predaceous. |
| * An asterisk means the genus or species is available commercially. aListed beneficials are indigenous, introduced into New Mexico or available from commercial sources. |
|
Common Beneficial Fly Families
Beneficial flies (Table 14) generally are small, two-winged, soft-bodied insects, which may be predaceous as adults or larvae. Tachinid flies are parasitoids as larvae. Some adult flies in this group, such as syrphids, feed on pollen and nectar. Eggs often are laid among prey, such as aphids. Larvae typically are conical and maggotlike in appearance.
The majority of these beneficial insects are common, native species with widespread distributions throughout North America.
Table 14. Common species of beneficial flies, with notes on their life cycles and habitats.
| Fly Family | Notes on Life Cycle, Habitats |
| Asilidae, robber flies | Asilids are common, generalist predators. The larvae are predaceous soil-dwellers. |
| Cecidomyiidae, gall midges, including some predaceous midges such as Aphidoletes sp.* | Most cecidomyiids are gall flies; however, larvae of several species are predators and parasitoids of aphids, scales and mites. Aphidoletes, attacks more than 60 aphid species and is used as an aphid predator in greenhouses. |
| Bombyliidae, bee flies | Bombyliidae are parasites of grasshopper eggs and the larvae of butterflies, moths, wasps and flies. Adults are large hairy flies that lay eggs close to hosts. Larvae hatch from the eggs and attack and enter the hosts. |
| Syrphidae, hover flies, Figures 53, 54 | Some syrphid fly larvae attack aphids, small caterpillars and perhaps thrips. Adults feed on pollen and nectar. Females lay single, white eggs near or among aphid colonies (Figures 43, 44). |
| Tachinidae | Tachinid flies attack immature beetles, butterflies and moths, earwigs, grasshoppers and true bugs. Eggs are laid on hosts or plants. Adult tachinids often resemble robust, hairy horseflies. Larvae are parasitoids in their hosts. |
Common Terrestrial Predatory Beetle Families
Beetles (Table 15) are the largest group of insects, including more than 110 families, many of which are predaceous. All beetles have four life stages: eggs, larvae, pupae and adults. Lady beetles belong to one of the most important groups of predaceous beetles in New Mexico. They may be host or nonhost specific, and they may be adapted for feeding in arborial or low-growing vegetation. Lady beetles may overwinter in the mountains (convergent lady beetles) or in local protected areas (ashgrey lady beetles). Lady beetles are available from commercial insectaries.
Table 15. Common families of predatory beetles and notes on their life cycles and habitats.a
| Beetle Family and Examples | Notes on Life Cycle, Habitats |
| Lady beetles, Coccinellidae 1. Aphid-feeding lady beetles Convergent lady beetles, Hippodamia convergens,* Figures 55, 56, 57. Ash gray lady beetle, Olla v-nigrum (arborial), Figure 58. Adalia bipunctata Asian lady beetle, Harmonia axyridis (introduced), Figures 59, 60. Seven spotted lady beetle, Coccinella septempunctata Twice stabbed lady beetle, Chilocorus stigma, Figure 61. 2. Mite-feeding lady beetles Stethorus picipes, S. punctum, S. punctillum 3. Whitefly-feeding lady beetles Delphastus*, Chilocorus, Hippodamia*, Clitostethus and Scymnus |
Most adults and larvae feed on soft-bodied insects or mites. Adults are dome-shaped, ranging from tiny to medium-sized, and from black to brightly colored. Larvae are active, elongate, have long legs, and resemble tiny alligators. About 500 species in the United States and Canada. |
| Soft winged flower beetles, Melyridae, Collops sp., Figure 62 | Collops beetles, Figure 62, feed on aphids, mite eggs and larvae of other arthropod species. Larvae are found in the soil. Common adults are dark blue, sometimes metallic, with orange-red markings. |
| Predaceous ground beetles, Carabidae | Ground beetles, primarily nocturnal. Variable in size. Most are predaceous as adults and larvae; they are usually black or dark colored and have long legs. Ground beetles have bean-shaped trochanters at the base of the hind leg not to be confused with similar darkling beetles which lack those bean-shaped trochanters (Figure 63). |
| *An asterisk means the genus or species is available commercially. aListed beneficials are indigenous, introduced into New Mexico or available from commercial sources. |
|
Common Groups of Parasitoids
Insect Egg Parasitoids
The egg parasitoids are a specialized group of tiny wasps that attack insect eggs. Several groups are present in New Mexico (Table 16). Trichogramma are the most commonly known. They probably are more habitat specific than host specific. Many different species can be purchased from commercial insectaries for use in controlling certain moth and butterfly populations. When ordering Trichogramma from commercial sources, ask for help in getting the correct species for a particular pest.
Table 16. Examples of some common parasitoids of insect eggs, with notes on hosts or habitats.a
| Parasitic species | Hosts or habitats |
| Anaphes iole | Field crop lygus bug eggs, e.g. cotton and alfalfa |
| Trichogramma minutum* | Tree crop moth eggs, e.g. fruit and nut trees |
| Trichogramma platneri* | Tree crop moth eggs, e.g. fruit and nut trees |
| Trichogramma pretiosum* | Field crop moth eggs, including vegetable and bedding plants |
| Trichogramma bactrae* | Field crop moth eggs, including vegetable and bedding plants |
| Trichogramma evanescens* | Field crop moth eggs, including vegetable and bedding plants |
| Trichogramma brassicae* | Field crop moth eggs, including vegetable and bedding plants |
| *An asterisk means the genus or species is available commercially. aListed beneficials are indigenous, introduced into New Mexico or available from commercial sources. |
|
Selected Families of Parasitic Wasps
There are more than 40 families of parasitic wasps, which include thousands of individual species. These parasitoids vary in size, shape and color and may be found in a variety of habitats. Insect parasitoids do not sting people. They kill their insect hosts in short order and may have a profound effect on host population density. Many species of parasitic wasps are available from commercial insectaries. Table 17 lists some of the more common parasitic wasps in New Mexico.
Table 17. Common families of parasitic wasps, with notes on their biology and hosts.a
| Common Families of Parasitic Wasps | Common Genera with Notes on Biology and Hosts |
| Aphelinidae (approximately 1,000 species) | Aphelinids attack mealybugs, whiteflies, scales and psyllids. Important genera include Aphelinus*, Aphytis*, Coccophagus, Encarsia*, Eretmocerus* and Prospaltella. Form black mummies (parasitized aphids). |
| Aphidiidae (approximately 1, 000 species) | Aphidiidae attack aphids. Important genera include Aphidius*, Lysiphlebus*, Praon, Diaeretiella*, Ephedrus, and Trioxys. Aphidiids form tan mummies, Figure 64. |
| Braconidae (approximately 1,000 species) | Braconids attack larvae of beetles, moths and butterflies, flies and sawflies. Important genera include Bracon, Chelonus, Cotesia (=Apanteles) and Opius. |
| Chalcididae (approximately 1,500 species) | Chalcidids are external or internal parasitoids of flies, butterflies, and moth larvae and pupae. Some attack beetles or are hyperparasites. |
| Encyrtidae (approximately 3,000 species) | Internal parasitoids of ticks, insect eggs, larvae and pupae of beetles, bugs, mealybugs and scales. Genera include Anagyrus, Comperiella Copidosoma, Encyrtus, Leptomastix, Metaphycus, Pentalitomastix and Psyllaephagus. |
| Eulophidae (approximately 3,400 species) | Eulophids are internal or external parasitoids of eggs, larvae or pupae of flies, moths and mites, scales, spiders and thrips. Genera include Aprostocetus, Chrysocharis, Diglyphus, Oomyzus (=Tetrastichus), Tamarixia. |
| Ichneumonidae (approximately 3,100 species) | Ichneumonids are external or internal parasitoids of beetles, butterflies, moths and wasp larvae or pupae. Genera include Bathyplectes, Campoplex, Diadegma, Exochus, Hyposoter, Ophion and Venturia. |
| Mymaridae (approximately 1,300 species) | Mymarids are internal parasites of insect eggs of beetles, flies grasshoppers, leafhoppers and true bugs. Genera include Anagrus* and Anaphes*. |
| Pteromalidae (approximately 3,000 species) | Pteromalids attack beetles, flies and other wasps. Genera include Dibrachys, Perilampus, Pteromalus and Scutellista. |
| Scelionidae (approximately 300 species) | Scelionids are internal parasitoids of spider, bug and moth eggs. Genera include Telenomus and Trissolcus. |
| Trichogrammatidae (approximately 650 species) | Trichogramma are parasitoids of externally placed insect eggs. |
| *An asterisk means the genus or species is available commercially. aListed beneficials are indigenous, introduced into New Mexico or available from commercial sources. |
|
Pathogens
The third category of beneficial organisms that may affect some yard and garden pests include pathogens. In short, pathogens are disease-causing organisms or agents, including bacteria, fungi, viruses and protozoa (single-celled organisms more complex than bacteria). These pathogens may occur naturally in an area or they may become prominent only when conditions allow. A few are available commercially to home gardeners (Table 18). While the typical hosts of these pathogens are various yard and garden pests, commercial formulations, where available, should be handled with the care given to pesticides. Read all labels on commercially available products and follow directions completely.
Pathogenic nematodes actually kill target insect pests by transmitting insect-pathogenic bacteria.
Table 18. Commercially Available Pathogens of Arthropods.
| Pest Controlled | Type | Pathogen Name |
| Japanese beetle larvab | B | Bacillus lentimorbus Bacillus popilliaec |
| Mosquito larvae | B | Bacillus sphaericus Bacillus thuringensis ssp. israelensis |
| Greater wax moth | B | Bacillus thuringiensis aizawai |
| Larvae of butterflies and moths | B | Bacillus thuringiensis ssp. kurstaki |
| Larvae of blackflies and fungus gnats | B | Bacillus thuringienis ssp. israelensis |
| Elm leaf beetle and Colorado potato beetle | B | Bacillus thuringiensis ssp. sandiego or tenebrionis |
| Aphids, crickets, grasshoppers, locusts, thrips, whiteflies, etc. | F | Beauveria bassiana |
| Beet armyworm larvae | V | Beet armyworm nuclear polyhedrosis virus (NPV) |
| Codling moth larvae | V | Codling moth granulosus virus |
| Bollworm | V-N | Heliothis NPV, Steinernema riobravisa |
| Flea beetle, Japanese beetle larvae, root maggots, grubs, etc. | N | Heterorhabditis bacteriophoraa |
| Mosquito larvae | F | Lagenidium giganteum |
| Cockroaches, flies | F | Metarhizium anisopliae |
| Grasshoppers, crickets, locusts | P | Nosema locustae |
| Aphids, whiteflies | F | Paecilomyces fumosoroseus |
| White grubs, Japanese beetle larvaeb, larvae of billbugs, flea beetles, and numerous other soil dwelling insects |
N | Steinernema carpocapsaea |
| Fungus gnat larvae and other soil dwelling insects | N | Steinernema feltiaea |
| Caterpillars | N | Steinernema glaseria |
| aNematodes kill by transmitting insect pathogenic bacteria. bJapanese beetle is currently known only from a limited area in Bernalillo county. cCommercial formulations of this bacterium perform poorly against other species of scarab beetles in New Mexico. Type: B, bacterium; F, fungus; V, virus; N, nematode; P, protozoan. |
||
References
“Suppliers of Beneficial Organisms in North America,” a comprehensive list of companies that sell natural enemies is available on the Web: www.cdpr.ca.gov/docs/pestmgt/ipminov/bensuppl.htm, or from the Web site sponsor, the California Department of Pesticide Regulation, Pest Management Analysis and Planning Program, 1020 N St., Rm 161, Sacramento, CA 95814-5604.
Photo Credits
Figures 1-20. Joe Ellington, New Mexico State University, Las Cruces
Figure 11. James Solomon, USDA Forest Service. www.insectimages.org
Figure 12. Clemson University, USDA Cooperative Extension Slide Series. www.insectimages.org
Figure 13. R.E. Rice, University of California Statewide IPM Project
Figure 14. Jack Kelly Clark, University of California Statewide IPM Project
Figure 15. Cooperative Extension Slide Series, Clemson University - USDA, www.insectimages.org
Figure 19. U. Jack Kelly Clark, California Statewide IPM Project
Figure 27. U. Jack Kelly Clark, California Statewide IPM Project
Figures 32-39. D. B. Richman, New Mexico State University, Las Cruces
Appendix A.
Pictures of Harmful and Beneficial Arthropods
 |
 |
| Figure 1. Spider mite egg and adult. | Figure 2. Western flower thrips. |
 |
 |
| Figure 3. Green peach aphids. | Figure 4. Yellow pecan aphid nymph. |
 |
 |
| Figure 5. Black margined pecan aphid nymph. | Figure 6. Black margined pecan aphid adult. |
 |
 |
| Figure 7. Black pecan aphid adult. | Figure 8. Black pecan aphid damage to pecan leaves. |
 |
 |
| Figure 9. Greenhouse whiteflies, nymphs and adults. | Figure 10. Cottony cushion scale. |
 |
 |
| Figure 11. Apricot scale. | Figure 12. Euonymus scale. |
 |
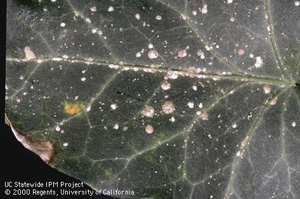 |
| Figure 13. San Jose scale. | Figure 14. Oleander scale. |
 |
 |
| Figure 15. Mealybugs. | Figure 16. Lygus bug nymph. |
 |
 |
| Figure 17. Lygus bug adult. | Figure 18. Stink bug adult. |
 |
 |
| Figure 19. Elm leaf beetle eggs, larva and adult. | Figure 20. Cabbage looper larva. |
 |
 |
| Figure 21. Beet armyworm larva. |
Figure 22. Pecan nut casebearer damage to young pecan nuts. Note bored holes, frass and fine silken webs. |
 |
 |
| Figure 23. Pecan nut casebearer larva. | Figure 24. Pecan nut casebearer pupa. |
 |
 |
| Figure 25. Pecan nut casebearer adult. | Figure 26. Sulfur butterfly larva. |
 |
 |
| Figure 27. Codling moth adult. | Figure 28. Tomato fruitworm egg. |
 |
 |
| Figure 29. Tomato fruitworm larva on pepper. | Figure 30. Tomato fruitworm pupae. |
 |
 |
| Figure 31. Tomato fruitworm adult. | Figure 32. Jumping spider. |
 |
 |
| Figure 33. Jumping spider. | Figure 34. Lynx spider. |
 |
 |
| Figure 35. Small wolf spider. | Figure 36. Orb-weaving spider. |
 |
 |
| Figure 37. Comb-footed spider. | Figure 38. Sac spider. |
 |
 |
| Figure 39. Crab spider. | Figure 40 Minute pirate bug nymph. |
 |
 |
| Figure 41. Minute pirate bug adults. | Figure 42. Big-eyed bug nymph. |
 |
 |
| Figure 43. Big-eyed bug adult | Figure 44. Damsel bug nymph. |
 |
 |
| Figure 45. Damsel bug adult. | Figure 46. Assassin bug eggs. |
 |
 |
| Figure 47. Assassin bug nymph. | Figure 48. Assassin bug adult. |
 |
 |
| Figure 49. A predatory mired bug, (Deraeocoris nebulosus). |
Figure 50. Green lacewing egg on stalk. |
 |
 |
| Figure 51. Green lacewing pupa. | Figure 52. Green lacewing adult. |
 |
 |
| Figure 53. Syrphid fly larva. | Figure 54. Syrphid fly adult. |
 |
 |
| Figure 55. Convergent lady beetle larva. | Figure 56. Adult convergent lady beetle. |
 |
 |
| Figure 57. Convergent lady beetle adults on a pecan tree trunk. |
Figure 58. Ash gray lady beetle adult. |
 |
 |
| Figure 59. Asian lady beetle larva. | Figure 60. Asian lady beetle adult. |
 |
 |
| Figure 61. Twice stabbed lady beetle adult. | Figure 62. Collops beetle adult. |
 |
 |
| Figure 63. Bean-shaped trochanter found on an adult ground beetle. The beetle on the right, a darkling beetle, lacks this feature. | Figure 64. Aphid mummy. Emergence of a minute parasitic wasp kills the aphid host, leaving a brown aphid skeleton with a hole in it. |
To find more resources for your business, home, or family, visit the College of Agricultural, Consumer and Environmental Sciences on the World Wide Web at pubs.nmsu.edu.
Contents of publications may be freely reproduced for educational purposes. All other rights reserved. For permission to use publications for other purposes, contact pubs@nmsu.edu or the authors listed on the publication.
New Mexico State University is an equal opportunity/affirmative action employer and educator. NMSU and the U.S. Department of Agriculture cooperating.
Printed and electronically distributed June 2005, Las Cruces, NM.
